Which alkyl halide and what conditions should be used to prepare the following alkene in good yield
Question:
Which alkyl halide and what conditions should be used to prepare the following alkene in good yield by an E2 elimination?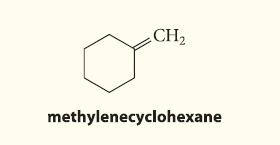
Transcribed Image Text:
CH₂ methylenecyclohexane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
If this alkene is to be produced in an E2 reaction from an alkyl halide the halide must be located a...View the full answer

Answered By

Muqadas Javed
I am a mentor by profession since seven years. I have been teaching on online forums and in universities. Teaching is my passion therefore i always try to find simple solution for complicated problems or task grasp them so that students can easily grasp them.I will provide you very detailed and self explanatory answers and that will help you to get good grade. I have two slogans: quality solution and on time delivery.
4.60+
24+ Reviews
144+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
What alkyl halide and what alkene would yield each of the following cyclopropane derivatives in the presence of a strong base? (a) Br Br (b) Ph H HC HC CH 3 CH3
-
Show how the Wittig reaction might be used to prepare the following alkenes. Identify the alkyl halide and the carbonyl components that would he used. (b) (a)
-
Financial statements for the ACTACTSTAT Company are given below: During 2021 the firm declared and paid cash dividends of P85,000. There were 50,000 shares of common stock outstanding throughout the...
-
GASB considers budgetary accounting and reporting to be important. List the principles outlined by GASB related to budgetary accounting and reporting.
-
A firm with a corporatewide debt-to-equity ratio of 1:2, an after-tax cost of debt of 7%, and a cost of equity capital of 15% is interested in pursuing a foreign project. The debt capacity of the...
-
FRAUD PREVENTION AND DETERRENCE IN ACTION Assume the following facts: thirty-day collection period. The invoice is used to post-sales to the accounting system. Checks are received in the mailroom and...
-
Worley Company buys surgical supplies from a variety of manufacturers and then resells and delivers these supplies to hundreds of hospitals. Worley sets its prices for all hospitals by marking up its...
-
Development economics studies the transformation of emerging nations into more prosperous one and it seeks to understand and shape the country's macro and microeconomics policies in order to lift...
-
What is the expected nucleophilic substitution product when (a) Methyl iodide reacts with Na + CH 3 CH 2 CH 2 CH 2 S ? (b) Ethyl iodide reacts with ammonia?
-
The crown ether [18]-crown-6 has a strong affinity for the methylammonium ion, CH 3 + NH 3 . Propose a structure for the complex between [18]-crown-6 and this ion. Show the important interactions...
-
Give the genus of the object. If the object has a genus larger than 5, write larger than 5.
-
Amit Krishnamurthy invests in a bond that promises him $6,000 each year for the next 20 years. If he pays $75,000 for this investment, what is his annual rate of return?
-
List and describe the business models of the three types of air cargo airlines.
-
Dillon Labs has asked its financial manager to measure the cost of each specific type of capital as well as the weighted average cost of capital (WACC). The WACC is to be measured by using the...
-
Beryls Iced Tea currently rents a bottling machine for $55,000 per year, including all maintenance expenses. It is considering purchasing a machine instead, and is comparing two options: a. Purchase...
-
How do the needs of air freight shippers differ from airline passengers for air transportation?
-
List at least three important behavioral issues related to the capital-budgeting process.
-
Where are the olfactory sensory neurons, and why is that site poorly suited for their job?
-
What product would expect to obtain from base treatment of 1,6-cyclo-decacedione? Base 1,6-Cyclodecanedione
-
Show the products you would except to obtain by claisen condensation of the following esters: (a) (CH3)2CHCH2COEt (b) Ethyl phenyl acetate (c) Ethyl cyclohexylacetate
-
As shown in figure 23.5, the Claisen reaction is reversible. That is, a ??keto ester can be cleared by base into two fragments. Using curved arrows to indicate electron flow, show the mechanism by...
-
The Bank of England's Monetary Policy Committee raised its interest rates fourteen times since December 2021. As in the US, firms in England often borrow funds or take funds out of alternative uses...
-
Eliminate Affordable Care Act subsidies for those earning between 300 and 400 percent of the federal poverty level. The Affordable Care Act (Obamacare) allows individuals and families to purchase...
-
The Moroccan monetary authority is using a heavily managed float to keep the dirham at $0.12 per dirham. Under current foreign exchange market conditions, nonofficial supply and demand would clear at...

Study smarter with the SolutionInn App


