Which of the following species should be aromatic by the Hckel 4n + 2 rule? (a) thiophene
Question:
Which of the following species should be aromatic by the Hückel 4n + 2 rule?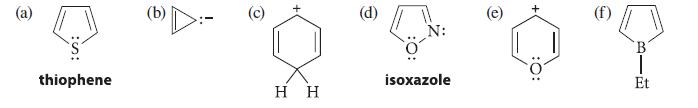
Transcribed Image Text:
(a) thiophene G O H H (d) N: isoxazole :0: B Et
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a b d Thiophene is aromatic One electron pair on sulfur is part of the aromatic 7electron system the ...View the full answer

Answered By

Evans Cherono
I am an Information Technology Graduate and willing to work on any computer science or IT work to ensure I do my best all the time.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the compounds or ions in Problem 15.38 are likely to be antiaromatic? Explain. Problem 15.38 Which of the following species should be aromatic by the Hckel 4n + 2 rule? (a) thiophene G O H H...
-
Which d the following species should be aromatic by the Huckel 4n + 2 rule? (a) (b) (c) N: isoxazole C2Hs
-
Which of the following species (there may be more than one) is are likely to have the structure shown here: (a) XeF4 (b) BrFe4+ (c) SiF4 (d) TeCI4 (e) HClO4? (The colors do not reflect atom...
-
Problem 5: Closing entries. (10 points) Given the following accounts, prepare the closing entries in the order and method taught using Income Summary. Number each entry 1, 2, .... Accounts payable $...
-
Give an example of a cost that can be directly matched with the revenue produced by an accounting firm from preparing a tax return.
-
Rhiannon Corporation has bonds on the market with 11.5 years to maturity, a YTM of 7.6 percent, and a current price of $1,060. The bonds make semiannual payments. What must the coupon rate be on...
-
Respondents Varsity Brands, Inc., Varsity Spirit Corporation, and Varsity Spirit Fashions & Supplies, Inc., design, make, and sell cheerleading uniforms. Respondents have obtained or acquired more...
-
Karla Tanner opens a Web consulting business called Linkworks and completes the following transactions in its first month of operations. April 1 Tanner invests $80,000 cash along with office...
-
Create an interface, Stock derivatives, with two attributes viz., delta=0.5 and vega=0.2 which are used to determine option premium of a stock. Also add an abstract method Find Premium. Write a class...
-
Using the theory of aromaticity, explain the finding that A and B are different compounds, but C and D are identical. (That A and B are different molecules was established by Prof. Barry Carpenter...
-
How many bonding MOs are there in a planar, cyclic, conjugated hydrocarbon that contains a ring of 10 carbon atoms? How many electrons does it have? How many of the electrons can be accommodated in...
-
On December 31, 2014, Hurly Co. performed environmental consulting services for Cascade Co. Cascade was short of cash, and Hurly Co. agreed to accept a $300,000 zero-interest-bearing note due...
-
In Figure 4, find equilibrium price and quantity (in dollars and units, respectively). Price ($) 12 10 8 6 4 2 Figure 4 S D 10 20 30 40 50 60 70 Quantity
-
Find two outside examples of how companies created new product lines as a result of identifying market opportunities or problems.
-
List three types of visuals and their purposes. How do visuals enhance marketing research reports?
-
What is the difference between consumers and customers? Why might the link to consumers be more important today?
-
What factors go into developing the right marketing strategy? How might the right marketing strategy change over time?
-
Helena has assets of $130,000 and liabilities of $160,000. One of her debts is for $120,000. Discuss the tax consequences of the reduction of this debt in each of the following circumstances: a. The...
-
After Theorem 1.5 we note that multiplying a row by 0 is not allowed because that could change a solution set. Give an example of a system with solution set S0 where after multiplying a row by 0 the...
-
Carvone is an unsaturated ketone responsible for the odor of spearmint. If carvone has M = 150 in its mass spectrum and contains three double bonds and one ring, what is its molecular formula?
-
Carvone (Problem 12.39) has an intense infrared absorption at 1690 cm1. What kind of ketone does carvone contain?
-
The (a) mass spectrum and the (b) infrared spectrum of an unknown hydrocarbon are shown. Propose as many structures as youcan. (a) 100 80 60 40 20 10 20 40 60 80 100 120 140 m/z (b) 60 40 20 - 4000...
-
On June 30, the end of the first year of operations, Johnson Industries, Inc., manufactured 3,700 units and sold 3,200 units. The following income statement was prepared, based on the variable...
-
The graph shows the tide level in Daytona Beach on a certain day. Time is measured in hours after midnight and the height of the tide is measured in feet. A) When was high tide? B) When was low tide?...
-
At the beginning of Year 2, the Redd Company had the following balances in its accounts: Cash $ 17,300 Inventory 7,500 Land 2,700 Common stock 16,000 Retained earnings 11,500 During Year 2, the...

Study smarter with the SolutionInn App


