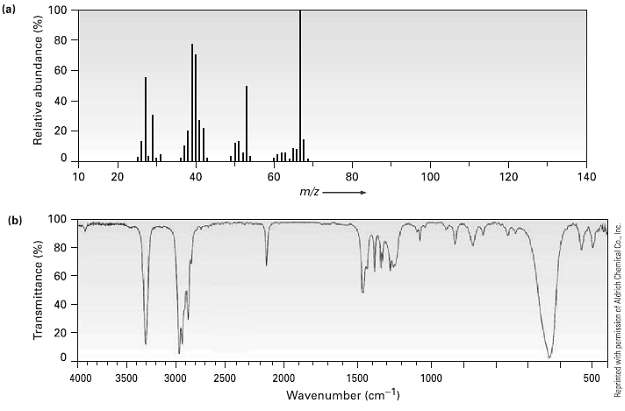
The (a) mass spectrum and the (b) infrared spectrum of an unknown hydrocarbon are shown. Propose as
Question:
The (a) mass spectrum and the (b) infrared spectrum of an unknown hydrocarbon are shown. Propose as many structures as youcan.
Transcribed Image Text:
(a) 100 80 60 40 20 10 20 40 60 80 100 120 140 m/z (b) 60 40 20 - 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-1) Transmittance (%) Relative abundance (%) Reprinted with permissicn et Aldrich Chemical Co. Inc.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The peak of maximum intensity base peak in the mass spectrum occurs at mz 67 This ...View the full answer

Answered By

Seema kuldeep
although I don't have an experience of teaching in a particular institute, previously I was an expert on Chegg and I have used to teach my batch mates and also my juniors.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The mass spectrum and infrared spectrum of an unknown compound are shown in Figures 13.27 and 13.28, respectively. Identify the compound. Figure 13.27 The mass spectrum for Problem 28. 100 E 80 3 60...
-
An unknown compound gives a mass spectrum with a weak molecular ion at m/z 113 and a prominent ion at m z 68. Its NMR and IR spectra are shown here. Determine the structure, and show how it is...
-
An unknown, foul-smelling hydrocarbon gives the mass spectrum and infrared spectrum shown. (a) Use the mass spectrum to propose a molecular formula. How many elements of unsaturation are there? (b)...
-
Reply as to whether you believe the following statements are correct (C) or incorrect (I) concerning PPS sampling. a. The size of a PPS sample is not based on the estimated variation of audited...
-
An article on price wars by two McKinsey consultants makes the following argument. That the (tit-for-tat) strategy is fraught with risk cannot be overemphasized. Your competitor may take an...
-
Table 10.3 shows that the van der Waals b parameter has units of L/mol. This means that we can calculate the sizes of atoms or molecules from the b parameter. Refer back to the discussion in Section...
-
What are the differences between project, program and portfolio management?
-
Determine Ending Accounts Receivable your accounts receivable clerk, Mary Herman, to whom you pay a salary of $1,500 per month, has just purchased a new Buick. You decided to test the accuracy of the...
-
Make a few paragraphs answering these questions about the movie Failure to launch (Please add detailed reasonings, Use the laws and rules we have covered throughout the semester and the facts in the...
-
A circuit youre using discharges a 20 F capacitor through an unknown resistor. After charging the capacitor, you close a switch at t = 0 s and then monitor the resistor current with an ammeter. Your...
-
Carvone (Problem 12.39) has an intense infrared absorption at 1690 cm1. What kind of ketone does carvone contain?
-
The (a) mass spectrum arid the (b) infrared spectrum of another unknown hydrocarbon is shown. Propose as many structures as youcan. (a) 100 80 60 40 20 20 60 80 100 120 140 10 40 m/z 100 (b) * 80 60...
-
Each plate of a parallel-plate air capacitor has an area S. What amount of work has to be performed to slowly increase the distance between the plates from xl to x2 if? (a) The capacitance of the...
-
A Campus Republicans fundraiser offers raffle tickets for $11 each. The prize for the raffle is a $300 television set, which must be purchased with the proceeds from the ticket sales. Find a function...
-
Using the fact pattern from assignment one (which is attached below), draft ten interrogatories (only four may come from the standard interrogatories under the South Carolina Rules of Civil...
-
A metal bowl with a weight of 1.10 N is placed in a larger kitchen container filled with coconut oil. How much coconut oil must the bowl displace in order to float? For reference, the mass density of...
-
David Brown was an Engineer in 1994 and earned $12,000 that year. His Daughter, Kelly Brown is an accountant and she earned $210,000 in 2018. The price index was 17.6 in 1994 and 218.4 in 2018. Kelly...
-
1. The focal length of a diverging lens is negative. If f = 16 cm for a particular diverging lens, where will the image be formed of an object located 45 cm to the left of the lens on the optical...
-
A sign is suspended by two wires as shown in Figure P10.12. Is the tension in each wire larger than, equal to, or smaller than the gravitational force exerted on the sign by Farth? Data from Figure...
-
1) Predict the organicproduct formed when BzCl reacts with cyclohexanol. BzCl = benzoylchloride. 2) Provide the majororganic product of the reaction below. 3) Draw the structureof the product formed...
-
The Safe Drinking Water Act (SDWA) sets a limit for mercurya toxin to the central nervous systemat 0.0020 ppm by mass. Water suppliers must periodically test their water to ensure that mercury levels...
-
Dimethylamine, (CH3)2NH has a molecular weight of 45 and a boiling point of 7.4 C. Trimethylamine, (CH3)3N has a higher molecular weight (59) but a lower boiling point (3.5 C). Explain this apparent...
-
Predict which member of each pair will be more acidic. Explain your answers. (a) Methanol or tert-butyl alcohol (b) 2-chloropropan-1-ol or 3-chloropropan-1-ol (c) 2-chloroethanol or...
-
Without looking them up, rank the following compounds in decreasing order of acidity. These examples represent large classes of compounds that differ widely in acidity. water, ethanol,...
-
1. Where are you on Maslow's hierarchy? Explain your answer. 2. Explain self-determination/cognitive evaluation theory (for this question, assume they're basically the same thing) giving a personal...
-
Select a participant (colleague, family member, or friend) that you would like to receive feedback from. Use the information from the table to plan a two-way, open, and evaluative feedback session as...
-
Imagine you are the assistant manager of the place in which you currently work. You have been instructed by the manager to organize and facilitate a company team building activity to promote...

Study smarter with the SolutionInn App


