Which of the molecules in each of the following pairs should have identical IR spectra, and which
Question:
Which of the molecules in each of the following pairs should have identical IR spectra, and which should have different IR spectra (if only slightly different)? Explain your reasoning carefully.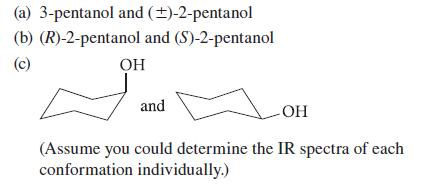
Transcribed Image Text:
(a) 3-pentanol and (±)-2-pentanol (b) (R)-2-pentanol and (S)-2-pentanol (c) OH and -OH (Assume you could determine the IR spectra of each conformation individually.)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The principle is that compounds that have different physical properties have diffe...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
If the NMR spectra of the molecules in Problem 30 were recorded by using high resolution for each nucleus, what differences would be observed?
-
Which of the molecules in Exercise 88 have net dipole moments (are polar)? SeO3 SeO2 PCl3 SCl2 SiF4
-
Which of the molecules in Exercises 91 and 92 have net dipole moments (are polar)? Exercise 91. a. XeCl2 b. ICl3 c. TeF4 d. PCl5 Exercise 92. a. ICl5 b. XeCl4 c. SeCl6
-
Net income Depreciation expense Accounts receivable increase (decrease) Inventory increase (decrease) Accounts payable increase (decrease) Accrued liabilities increase (decrease) O Changes in current...
-
What are the purpose and importance of a procedures manual?
-
Cole Petroleum has spent $ 206,000 to refine 63,000 gallons of petroleum distillate, which can be sold for $ 6.30 per gallon. Alternatively, Cole can process the distillate further and produce 53,000...
-
Which interest rate on a bond determines the amount of the semiannual interest payment? a. Market rate b. Effective rate c. Stated rate d. None of the above
-
Refer to Problem 7.3. The head of the Mariposa County Accounting Department wants to establish a standard regarding the length of time that employees can expect to wait to receive reimbursement for...
-
The increasing of Halal food and cosmetics demand in South Korea has opened opportunities to Muslim country companies to expand their business in South Korea. Assuming you are working as a manager...
-
List the factors that determine the wavenumber of an infrared absorption.
-
The mass spectrum of 2-chloropentane shows large and almost equally intense peaks at m/z = 71 and m/z = 70. (a) Classify each peak as an even-electron or odd-electron ion. (b) What stable neutral...
-
Summarize the key advantages of stored procedures.
-
Classify the gears and explain them with neat sketches.
-
Describe the primary caretaker presumption and explain how the courts determine the identity of a childs primary caretaker.
-
Explain what a gender-neutral standard is in the context of custody decisions. Do you believe that custody decisions are, in fact, gender neutral? Why or why not?
-
What for Thermit welding is used.
-
Montgomery Company purchased an electric wax melter on April 30, 2016, by trading in its old gas model and paying the balance in cash. The following data relate to the purchase. Instructions Prepare...
-
What are the objectives of participative budgeting?
-
Reichenbach Co., organized in 2018, has set up a single account for all intangible assets. The following summary discloses the debit entries that have been recorded during 2018 and 2019. Instructions...
-
Without looking at Table 24.2, rank the following compounds in order of ascending basicity. (a) p-Nitro aniline, p-aminobenzaldehyde, p-Bromoaniline (b) p-Chloroanilinc, p-aminoacetophenone, p-methyl...
-
Calculate the percentages of neutral and protonated forms present in a solution of 0.0010 M pyrimidine at pH = 7.3. The pKa of pyrimidinium ion is 1.3.
-
Propose structures for either a nitrile or an amide that might be a precursor of each of the following amines: (a) CH3CH2CH2NH2 (b) (CH3CH2CH2)2NH (c) Benzyl amine, C6H5C2NH2 (d) N-Ethyl aniline
-
Imagine a scenario where Country A is a major steel producer and Country B is a developing nation with a growing steel industry. Country B's steel industry is facing significant challenges due to...
-
The following data, expressed as $10 4 , are the closed ledger accounts showing assets, liabilities, income, and expenses of Blue River Fabricators, Inc., a family owned firm that does not declare...
-
Use the figure below of indifference curves to answer the following questions. Assume that Curve B represents a total combined utility of 14 utiles (units of satisfaction) and that Curve C represents...

Study smarter with the SolutionInn App


