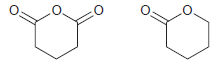
A 1 H NMR spectrum was acquired for each of the following two compounds. One spectrum exhibits
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
Only one signal downfield ...View the full answer

Answered By

Archana P M
My tutoring experience started when i was in high school. I used to teach topics like Physics and Mathematics to kids in lower grades. This continued in my college years also. Used to actively give talks in topics of Computer Science and Mathematics in various seminars organized in out college. Tutoring is a rewarding experience as I also get to learn new ways of thinking and solving problems from the people I teach.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following compounds and ions, 1. Draw a Lewis structure. 2. Show the kinds of orbitals that overlap to form each bond. 3. Give approximate bond angles around each atom except...
-
For each of the following compounds determine which (if any) lone pairs are participating in aromaticity. a. b. c. d. e. f. g. h. :N-H
-
For each of the following payroll-related taxes, indicate whether it generally applies to (1) Employees only, (2) Employers only, or (3) Both employees and employers: a. Federal income tax b. Federal...
-
What are two important limitations of the Heckscher- Ohlin theory?
-
Is there a preferred habitat for banks? credit unions? property insurance companies? life insurance companies?
-
Briefly explain why it is useful for accountants to understand a range of ethical perspectives despite having a professional code of ethics.
-
The Maxwell's relation \(\left(\frac{\partial T}{\partial V} ight)_{S}=-\left(\frac{\partial P}{\partial S} ight)_{V}\) is derived from the fundamental thermodynamic relation (a) \(d H=T d S+V d P\)...
-
In the Deep Creek Mining Company example described in this chapter Table, suppose again that labor is the variable input and capital is the fixed input. Specifically, assume that the firm owns a...
-
A firm has to pay 6 million euros forsupplies it recently received from Germany. Today, the euro hasappreciated by 3 percent against the U.S. dollar. The firm hasdetermined that whenever the euro...
-
The following table shows the percentage of on-time arrivals, the number of mishandled baggage reports per 1000 passengers, and the number of customer complaints per 1000 passengers for ten airlines...
-
On January 2, 2007, a Sunny Communications $1,000 face value, six-year bond sold for $889. Investors who bought this particular bond will be paid interest equal to $40 every six months. Market...
-
Firm As management is very conservative whereas Firm Bs is more aggressive. Is it true that, other things the same, Firm B would probably have larger holdings of marketable securities? Explain.
-
A solid uniform cone, of base radius 2a and height 5a, is suspended by a point, B, on the rim of its circular base. The centre of the circular base is denoted by C. Find the angle BC makes with the...
-
Here is where you will use your research on Ansoff's theory to help distinguish between strategic, administrative, and operational decisions from the perspective of a manager and a leader. Be sure to...
-
Alibaba's strategy and logistics to develop a unique business model allow me to see the takeaways that I learned that can help improve other companies' models. For example, you can create an...
-
The Buddhist tradition has a doctrine that views human beings as having No Self (Anatta). Explain the No Self Doctrine and how it differs from the Hindu view of the Atman. The Buddha sees that the...
-
Describe a situation in which you experienced two decision-making pitfalls. What techniques could be applied in each situation for more effective decision-making? Which decision-making techniques or...
-
When considering a change, which role would be responsible for analyzing the schedule, budget, and resource allocations to determine how the change impacts the project? Explain.
-
Perform the operation mentally. 154 54
-
2. Assume a person bends forward to lift a load "with his back" as shown in Figure P12.42a. The person's spine piv- ots mainly at the fifth lumbar vertebra, with the principal supporting force...
-
Which conformation of 1, 2-dibrornoethane (Problem 3.44) would you expect to have the larger dipole moment? The observed dipole moment of 1, 2-dibromoethane is = 1.0 D. What does this tell you about...
-
The barrier to rotation about the CC bond in bromoethane is 15kJ/mol (3.6kcal/mol). (a) What energy value can you assign to an HBr eclipsing interaction? (b) Construct a quantitative diagram of...
-
Draw the most stable conformation of pentane, using wedges and dashes to represent bonds corning out of the paper and going behind the paper, respectively.
-
List and analyze the selection to demonstrate whether you agree or disagree with the list. Explain why or why not....
-
Discuss how data deduplication works in backup systems and its impact on storage efficiency. What are some potential drawbacks of deduplication, and how might they be mitigated ?
-
Describe the importance of application consistency in backup operations. How do modern backup systems ensure consistency, and what are the risks of a quiescence failure ?

Study smarter with the SolutionInn App


