For each of the following compounds determine which (if any) lone pairs are participating in aromaticity. a.
Question:
a.
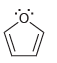
b.
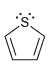
c.
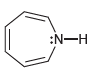
d.
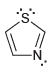
e.
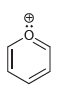
f.
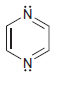
g.
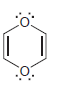
h.
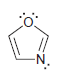
Transcribed Image Text:
:N-H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
a One of the lone pairs on oxygen b One of the lone pairs on s...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following compounds, determine whether or not you would expect its IR spectrum to exhibit a signal to the left of 3000 cm -1 a. b. c. . d. e. f.
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) OMe . CI H,
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) Discuss. OMe . CI H,
-
State Newtons second law of motion. What are the limitations on the use of Newtons second law? Explain.
-
The New England Cycle Company is planning next month's production of its two bicycles: its B2 tandem cycle built for two and its S10 single-rider 10-speed model. Both models use the same seats and...
-
Consider a one-dimensional system with potential \(V(x)=V_{0} /\left(x^{2}+a^{2} ight)\). Write the WKB approximation for the wave function for scattering, for energy \(0
-
For each of the following situations, calculate the degrees of freedom \((d f)\), identify the critical values (assume \(\mathrm{a}=.05\) [two-tailed]), calculate the standard error of the mean \(s\)...
-
In Figure where V23 = 3.00V1, n moles of a diatomic ideal gas are taken through the cycle with the molecules rotating but not oscillating. What are? (a) p2/p1 (b) p3/p1 and (c) T3/T1 For path 1 ?? 2,...
-
The Claussens are considering the purchase of a hardware store. The Claussens anticipate that the store will generate cash flows of $ 8 4 , 0 0 0 per year for 2 0 years. At the end of 2 0 years, they...
-
The term persuasion encompasses a suite of commercial and corporate brand communication activities notably marketing, advertising, branding, and public relations (together with public affairs) ...
-
Identify which of the following compounds is more acidic and explain your choice.
-
Go to the beginning of Section 18.1 where 8 best-selling drugs were shown. Review the structures of those compounds, and identify all of the aromatic rings that are not already highlighted in red. ...
-
The first term of a geometric progression is 1 and the second term is 2 sin x where /2 < x < /2. Find the set of values of x for which this progression convergent.
-
Assume that each consumer in Will's town has the following yearly demand for wine: Q120-2P. The cost to produce wine is C=150+10Q. Will wants to start a wine club with an annual membership fee and a...
-
Consider a duopoly market with only two sellers selling an identical product. Market demand is given by P = 40 - 1- Q. Technology in the industry is such that the marginal (and average) cost of...
-
Martex Inc. has 560,000 shares outstanding. Information on their issuance is as follows: Date January, 2022 June, 2023 Totals Shares Issue Issued Price Total Proceeds 400.000 $16.00 $6,400,000...
-
A special event that is considered incidental generated $470,000 in gross revenues and had direct costs of $350,000 resulting in net cash of $120,000 received. (Note: Record the revenues to Special...
-
Reflect on your readings and the Biblical Perspective as you think through the following questions: Is it wrong for a business to seek to maximize profits? What does the Bible say about earning...
-
Assume that the spinner cannot land on a line. Determine the probability that the spinner lands on (a) Red. (b) Green. (c) Yellow. (d) Not yellow.
-
1. Using the information from Problem 16-4B, prepare a statement of cash flows for Lim Garden Supplies Inc. using the direct method of presenting cash flows from operating activities. 2. How does Lim...
-
Would you expect the reaction of propyl bromide with sodium cyanide (NaCN), that is, CH3CH2CH2Br + NaCN CH3CH2CH2CN + NaBr to occur faster in DMF or in ethanol? Explain your answer.
-
When tert-butyl bromide undergoes solvolysis in a mixture of methanol and water, the rate of solvolysis (measured by the rate at which bromide ions form in the mixture) increases when the percentage...
-
List the following compounds in order of decreasing reactivity toward CH3O- in an SN2 reaction carried out in CH3OH: CH3F, CH3Cl, CH3Br, CH3I, CH3OSO2CF3, 14CH3OH.
-
3. Create a chart for gross margin less R&D and capital costs calculations. a. Calculate gross margin less R&D and capital costs for options A and B. b. Graph total gross margin less R&D and capital...
-
Why was Thomas Paine's "common sense" article significant to the american revolution?
-
Aramex, Inc is currently offering a bond with a par value of $1,000, which is priced at $1,488 in the market. The bond offers a fixed annual coupon rate of 14.05 percent, which is paid semiannually....

Study smarter with the SolutionInn App


