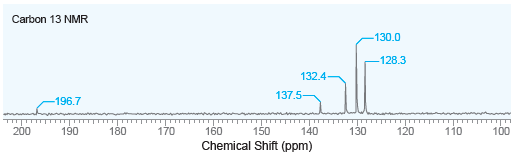
A compound with molecular formula C 13 H 10 O produces a strong signal at 1660 cm
Question:
Transcribed Image Text:
Carbon 13 NMR -130.0 -128.3 132.4- 137.5- -196.7 200 180 170 160 150 Chemical Shift (ppm) 140 130 120 110 100 190
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)

Answered By

Mario Alvarez
I teach Statistics and Probability for students of my university ( Univerisity Centroamerican Jose Simeon Canas) in my free time and when students ask for me, I prepare and teach students that are in courses of Statistics and Probability. Also I teach students of the University Francisco Gavidia and Universidad of El Salvador that need help in some topics about Statistics, Probability, Math, Calculus. I love teaching Statistics and Probability! Why me?
** I have experience in Statistics and Probability topics for middle school, high school and university.
** I always want to share my knowledge with my students and have a great relationship with them.
** I have experience working with students online.
** I am very patient with my students and highly committed with them
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A compound with molecular formula C 9 H 10 O exhibits the following spectra ( 1 H NMR, 13 C NMR, and IR). Identify the structure of this compound.. Proton NMR 10 Chemical Shift (ppm) Carbon NMR 128.5...
-
The IR spectrum of a compound with molecular formula C5H8O was obtained in CCl4 and is shown in Figure 13.42. Identify the compound. Wavelenga qum) 15 16 14 3600 340) 3800 3300 3000 280K 2600 2400...
-
A compound with molecular formula C 8 H 10 O produces six signals in its 13 C NMR spectrum and exhibits the following 1 H NMR spectrum. Deduce the structure of the compound. Proton NMR Chemical Shift...
-
The table shows the fees for refund anticipation loans (RALs) offered by an online tax preparation firm. Find the annual rate of interest for each of the following loans. Assume a 360-day year. (A) A...
-
Pete is considering placing a bet on the NCAA playoff game between Indiana and Purdue. Without any further information, he believes that each team has an equal chance to win. If he wins the bet, he...
-
A particular cylindrical Gaussian surface encloses no charge. If 20 electric field lines pass into the cylinder through one of the flat ends of the cylinder and 16 of these lines emerge from the...
-
Prepare shadow maps for point \(\mathrm{P}\) on the sun-path diagrams for \(35^{\circ} \mathrm{N}\) and \(40^{\circ} \mathrm{N}\) for the following three geometries shown in a, b, and c. Determine...
-
A local restaurateur who had been running a profitable business for many years recently purchased a three-way liquor license. This license gives the owner the legal right to sell beer, wine, and...
-
A power station generates electricity at 25,000 V, which is then stepped up to 500,000 V using a transformer for efficient transmission. If the primary coil has 200 turns, how many turns does the...
-
You are given a series of trades. Each trade has a key, a value, a quantity, and a sequence number. For each trade, print the weighted moving average of all the trades for that particular key. Round...
-
A compound with molecular formula C 9 H 10 O exhibits a strong signal at 1687 cm 1 in its IR spectrum. The 1 H and 13 C NMR spectra for this compound are shown below. Identify the structure of this...
-
A ketone with molecular formula C 9 H 18 O exhibits only one signal in its 1 H NMR spectrum. Provide a systematic (IUPAC) name for this compound.
-
Florin, owner of Florin's Flower Mart, randomly sampled 124 customers in order to obtain data such as a customer's method of payment (Visa, MasterCard, American Express, cash, or some other method)...
-
Harry Daffodil is a 3 rd year accounting student at Kwantlen and also works part - time as a bookkeeper for a plant store in Surrey. He would like you to prepare his 2 0 2 1 tax return and has...
-
Discover and report what products are sold and how they are related to the chiropractor or servic?
-
Management is confident that the company can sell 54,450 games next year (an increase of 9,450 games, or 21%, over last year). Given this assumption: a. What is the expected percentage increase in...
-
if a company that sells 3 0 million in merchandise every year estimates that about 1 million in merchandise will be returned for refund how should this revenue be recognized and reported ?
-
Explain how using "hold to maturity accounting" for SpinCo's assets affects the value of SpinCo. Is the market value that investors will pay for SpinCo different if the initial transfer of assets is...
-
Solve the problem. Find all values of b such that the straight line 3x - y = b touches the circle x 2 + y 2 = 25 at only one point.
-
Recall that Chapter 8 described the binary search algorithm for finding a particular entry in an ordered list. The idea behind binary search is to begin looking in the exact center of the list. If...
-
Rank the following alcohols in order of increasing ease of acid-catalyzed dehydration. OH OH OH
-
Acid-catalyzed dehydration of neopentyl alcohol, (CH3)3CCH2OH, yields 2-methyl-2- butene as the major product. Outline a mechanism showing all steps in its formation.
-
Acid-catalyzed dehydration of either 2-methyl-1-butanol or 3-methyl-1-butanol gives 2-methyl-2-butene as the major product. Write plausible mechanisms that explain these results.
-
In eukaryotes, which two processes occur during termination of transcription?
-
Below is the list of eukaryotic organelles. Mitochondria | Plant chloroplast | Nucleus | Lysosome | Nuclear envelope | Rough endoplasmic reticulum | Golgi apparatus | Smooth endoplasmic reticulum |...
-
Study customer touchpoints. Remember that we are working with the B2C market. Select two stages of the customer decision journey Answer the following questions: For each selected stage, identify a...

Study smarter with the SolutionInn App


