A Friedel-Crafts acylation is an electrophilic aromatic substitution in which the electrophile (E + ) is an
Question:
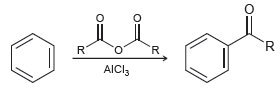
Transcribed Image Text:
R. AICI,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
00 J R AIC...View the full answer

Answered By

Cyrus Sandoval
I a web and systems developer with a vast array of knowledge in many different front end and back end languages, responsive frameworks, databases, and best code practices. My objective is simply to be the best web developer that i can be and to contribute to the technology industry all that i know and i can do. My skills include:
- Front end languages: css, HTML, Javascript, XML
- Frameworks: Angular, Jquery, Bootstrap, Jasmine, Mocha
- Back End Languages: Java, Javascript, PHP,kotlin
- Databases: MySQL, PostegreSQL, Mongo, Cassandra
- Tools: Atom, Aptana, Eclipse, Android Studio, Notepad++, Netbeans.
Having a degree in Computer Science enabled me to deeply learn most of the things regarding programming, and i believe that my understanding of problem solving and complex algorithms are also skills that have and will continue to contribute to my overall success as a developer.
I’ve worked on countless freelance projects and have been involved with a handful of notable startups. Also while freelancing I was involved in doing other IT tasks requiring the use of computers from working with data, content creation and transcription.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which compound will undergo an electrophilic aromatic substitution reaction more rapidly, benzene or hexadeuteriobenzene? or D- H.
-
A Friedel-Crafts alkylation is an electrophilic aromatic substitution in which the electrophile (E + ) is a carbocation. In previous chapters, we have seen other methods of forming carbocations, such...
-
For each compound below, identify which position(s) is/are most likely to undergo an electrophilic aromatic substitution reaction. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH3 NO2 O,N. CH3 O,N NO2
-
The United Arab Emirates (UAE) is unique in that expatriates constitute more than 80 per cent of the population. As the country continues to grow and accept foreigners at an astounding pace, Emiratis...
-
Explain the techniques for a small service firm setting an hourly price.
-
Larrys Lawn Equipment Company gives terms of 2/10, n/30. Larry has annual credit sales of $500,000 and average accounts receivable of $60,000. a. What is Larrys accounts receivable turnover? b. What...
-
Consider the population of 41 whole numbers from 0 to 40. What is the average of these numbers? Select three random samples of seven numbers from this list. Find the average of each sample. Compare...
-
Harte Systems, Inc., a maker of electronic surveillance equipment, is considering selling to a well-known hardware chain the rights to market its home security system. The proposed deal calls for the...
-
On May 1, Soriano Company reported the following account balances along with their estimated fair values: Receivables Inventory Copyrights Account Patented technology Total assets Current liabilities...
-
The assignment requires access to Westlaw online. At the West search box enter kc: 131 S. Ct. 2705 (Bullcoming v. New Mexico, 131 S. Ct. 2705 (2011)). What is the citation for the 2011 Illinois Court...
-
The following compound cannot be made with either a Friedel-Crafts alkylation or acylation. Explain.
-
Draw the two major products obtained when toluene undergoes monobromination.
-
Compare welfare in a market where a firm is both a monopsony and a monopoly to welfare in markets in which the firm has a monopsony in the input market but acts as a price taker in the output market.
-
Fowler has developed a number of approaches to reusability since his initial work on design patterns, Research some of his recent work on the theme of reusability: Select one and write a critical...
-
Use game theory to describe how an effort by two universities to recruit a top basketball player might result in both committing NCAA recruiting violations.
-
The presence of biophysical limits to economic growth from the perspective of ecological economics is understood through the following three salient features: - The scale effect (i.e., the sheer size...
-
Assume that you are the newly appointed head of the NCAA. Construct a frame- work for a new scholarship program that would entirely replace the existing system. Describe what should be its...
-
What is the elasticity of demand for season ski passes? When the price of a season ski pass is $400, buyers, whose demand curve for passes is linear, wish to purchase 10,000 passes per year, but when...
-
Solve each problem. Let a be directly proportional to m and n 2 , and inversely proportional to y 3 . If a = 9 when m = 4, n = 9, and y = 3, find a when m = 6, n = 2, and y = 5.
-
The following selected information was taken from Sun Valley Citys general fund statement of revenues, expenditures, and changes in fund balance for the year ended December 31, 2019: Revenues:...
-
Explain how you could differentiate between the compounds in each of the following pairs by using simple physical or chemical tests that give readily observable results, such as obvious solubility...
-
Explain how you could differentiate between the compounds in each of the following pairs by using simple physical or chemical tests that give readily observable results, such as obvious solubility...
-
Explain why a mixture of two isomeric ethers is formed in the following reaction. NaBH
-
American X wishes to borrow U.S. dollars at a fixed rate of interest. Corporation Y wishes to borrow Japanese Yen at a fixed rate of interest. The amounts required by the two companies are the same...
-
Bunga Raya Kuning Berhad is a Malaysian-based MNC that obtains 12 percent of its supplies from the U.K. manufacturers. Sixty-five percent of its revenues are from due to exports to U.K. where its...
-
Trevor's Tool Shop is considering investing in a new machine. The company currently has $500,000 per year in sales. The company has $265,000 per year in net income. If the company invests in the new...

Study smarter with the SolutionInn App


