For each compound below, identify which position(s) is/are most likely to undergo an electrophilic aromatic substitution reaction.
Question:
(a)
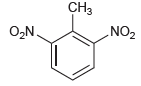
(b)
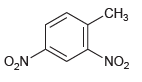
(c)
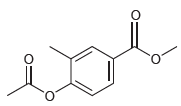
(d)
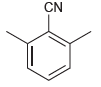
(e)
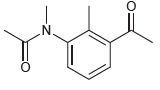
(f)
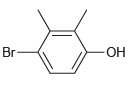
(g)
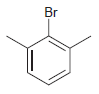
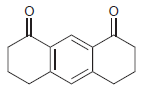
(h)
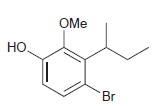
(i)
Transcribed Image Text:
CH3 „NO2 O,N. CH3 O,N NO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
a b c...View the full answer

Answered By

Antony Sang
I am a research and academic writer whose work is outstanding. I always have my customer's interests at heart. Time is an important factor in our day to day life so I am always time conscious. Plagiarism has never been my thing whatsoever. I give best Research Papers, Computer science and IT papers, Lab reports, Law, programming, Term papers, English and literature, History, Math, Accounting, Business Studies, Finance, Economics, Business Management, Chemistry, Biology, Physics, Anthropology, Sociology, Psychology, Nutrition, Creative Writing, Health Care, Nursing, and Articles.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each compound below, identify the most acidic proton in the compound: (a) (b) (c) (d) (e) (f) (g) (h) -NH2 -
-
For each compound below, identify all lone pairs and indicate whether each lone pair is localized or delocalized. Then, use that information to determine the hybridization state and geometry for each...
-
For each compound below, identify any polar covalent bonds, and indicate the direction of the dipole moment using the symbols + and -: a. HBr b. HCl c. H 2 O d. CH 4 O
-
The following two-dimensional problems all have mixed boundary conditions involving both traction and displacement specifications. Using various field equations, formulate all boundary conditions for...
-
What techniques can small business owners use to stretch their advertising budgets?
-
What does it mean to give legal advice?
-
Tourism Use a time series chart to display the data shown in the table. The data represent the percentages of Egypts gross domestic product (GDP) that come from the travel and tourism sector....
-
Inventoriable Costs George Solti, the controller for Garrison Lumber Company, has recently hired you as assistant controller. He wishes to determine your expertise in the area of inventory accounting...
-
"How can an organization leverage automated backup verification processes to ensure the reliability and recoverability of backup data? Discuss the potential role of machine learning and artificial...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
The following compound has four aromatic rings. Rank them in terms of increasing reactivity toward electrophilic aromatic substitution.
-
Predict the product(s) for each of the following reactions: (a) (b) (c) OH HNO, H,SO, OMe Br Br2 FeBr3
-
How would the effective annual interest rate for Problem 28 change if the bank charged $1,000 in closing costs? In Problem 28, determine the effective annual interest rate on a $100,000 short-term...
-
Follow up with the contact at the government department. Did they have any questions? Any comments on your work? Does the prototype do everything they wanted and needed? Does it work on their network...
-
Describe the externalities associated with a football stadium compared with an amusement park. Which would have greater positive externalities? Which would have greater negative externalities?
-
Select an application that is used in your organization or school and that has more than one user interface screen. Also, try to select an application that you haven't used before, or at least one...
-
The authority an agent reasonably assumes he or she has that relates to the express authority granted by the principal. a. agent b. agency by necessity c. agency by ratifi cation d. apparent a...
-
Indicate whether each of the following statements is true or false by writing T or F in t he a nswer c olumn. If an agency exists at will, the principal has the right to revoke the agency agreement...
-
Sketch the graph of an appropriate function and then use the graph to solve each equation or inequality. First determine whether the expression is an equation or an inequality. When appropriate, use...
-
The packaging division of a company having considered several alternative package designs for the company's new product has finally brought down their choices to two designs of which only one has to...
-
Calculate the energy in kJ mol-1 of the light described in Problem 12.1(b) Blue light with = 4800 A
-
The mass spectrum of methyl isobutyl ether does not show a peak due to inductive cleavage, in contrast to the mass spectrum of di-sec-butyl ether (Eq. 12.31). Use what you know about carbocation...
-
The mass spectrum of methyl isobutyl ether does not show a peak due to inductive cleavage, in contrast to the mass spectrum of di-sec-butyl ether (Eq. 12.31). Use what you know about carbocation...
-
A 0.012-kg bullet, traveling at 850 m/s, hits a 2-kg block of wood that is initially at rest, and goes straight through it. Assume that the final velocity of the bullet relative to the block is 400...
-
Assume that Iowa Co. will receive 1,200,000 Mexican pesos in 180 days. The Mexican interest rate is 3% over 180 days and the US rate is 2% over the same period. The spot rate for the Mexican peso is...
-
After watching the video from "The Big Bang Theory, describe what was occurring in the scene. What types of listening were demonstrated. How could this conversation be improved so that both parties...

Study smarter with the SolutionInn App


