Arrange each set of isomeric alkenes in order of stability. a. b.
Question:
a.
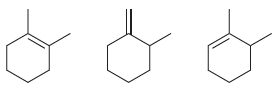
b.
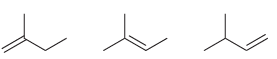
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
a b di substit...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Acid-catalyzed dehydration of 2,2-dimethyl-1-hexanol gave a number of isomeric alkenes including 2-methyl-2-heptene as shown in the following formula. (a) Write a stepwise mechanism for the formation...
-
Using only the periodic table, arrange each set of atoms in order from largest to smallest: (a) K, Li, Cs; (b) Pb, Sn, Si; (c) F, O, N
-
Using only the periodic table, arrange each set of atoms in order of increasing radius: (a) Ba, Ca,Na; (b) Sn, Sb, As; (c) Al, Be, Si.
-
PLEASE, PLEASE use an Excel formula:1. Begin the formula with an = sign.2. Reference cells, instead of entering values.Example: =PV(H8+H9) A company borrowed money from a local bank. The note the...
-
Comment on the validity of the results of Atterberg limits on soils G and H.>
-
1. Jim Adams posts the column totals and individual transactions in "Other" columns to the appropriate general ledger account and updates each general ledger account balance. He places a posting...
-
The function of a condenser is to (a) condense steam (b) maintain pressure below atmospheric pressure (c) increase work output of prime mover (d) all of the above
-
The following transactions apply to Walnut Enterprises for 2016, its first year of operations: 1. Received $50,000 cash from the issue of a short-term note with a 6 percent interest rate and a...
-
(1 point) A cryptography student has accidentally used the same keystream to encrypt two different messages, M and M2. We know that the ciphertexts are C 0x06c6d00e4633400a and C2 =...
-
Based on your answer to question 9, construct a first-quarter 2015cash flow forecast for Westmark Industrial. Westmark Industrials annual income statement and balance sheet for December 31, 2014...
-
Why is chlorine added to drinking water? What is the potential problem with adding chlorine to drinking water?
-
How do maximum containment levels and national emission limitations differ?
-
On February 1, 2017, Jura Corp. (Jura) issued an $8,000,000 bond with a 7 percent coupon rate and a maturity date of January 31, 2023. Interest is paid annually on January 31. The effective interest...
-
The current price of Estelle Corporation stock is $25. In each of the next two years, this stock price will either go up by 24% or go down by 24%. The stock pays no dividends. The one-year risk-free...
-
The Amazon.com website originally sold books; then the management of the company decided to extend their Web-based system to include other products. How would you have assessed the feasibility of...
-
The Saban Corporation is trying to decide whether to switch to a bank that will accommodate electronic funds transfers from Sabans customers. Sabans financial manager believes the new system would...
-
Harbin Manufacturing has 10 million shares outstanding with a current share price of $23.58 per share. In one year, the share price is equally likely to be $28 or $20. The risk-free interest rate is...
-
The Fast Reader Company supplies bulletin board services to numerous hotel chains nationwide. The owner of the firm is investigating the benefit of employing a billing firm to do her billing and...
-
Find the most general antiderivative of the function. f(x) = /2
-
Identify the most stable compound:
-
Ribose, an essential part of ribonucleic acid (RNA), has the following structure: (a) How many chirality centers does ribose have? Identify them. (b) How many stereo isomers of ribose are there? (c)...
-
On catalytic hydrogenation over a platinum catalyst, ribose (Problem 9.57) is converted into ribitol. Is ribitol optically active or inactive?Explain. CH2 Ribitol
-
Hydroxylation of cis-2-hutene with OsO4 yields 2, 3-butanediol. What stereochemistry do you expect for the product?
-
In this assignment, students must select a publicly listed entity, perform fundamental analysis and provide investment recommendations. The selected company can be one of the companies in your...
-
The following is an interesting interview with Ray Kurzweil that explains what the "Singularity" is. It is only two months ago, before the explosion Chat GPT....
-
When is a situation where a larger, more complex firm might have net income higher or lower than changes in cash (in other words, provide a concrete hypothetical setting where cash and net income...

Study smarter with the SolutionInn App


