Below are NMR spectra of several compounds. Identify whether these compounds are likely to contain ethyl, isopropyl,
Question:
(a)
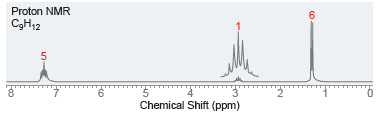
(b)
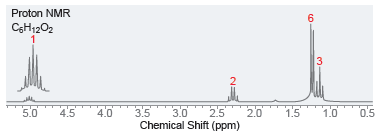
(c)
(d)
Transcribed Image Text:
Proton NMR C3H12 Chemical Shift (ppm) Proton NMR CgH12O2 3 5.0 4.5 4.0 3.5 2.0 3.0 Chemical Shift (ppm) 1.5 1.0 0.5 25
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
a The spectrum exhibits the characteristic pattern of an isopropyl group b The ...View the full answer

Answered By

Amit Kumar
I am a student at IIT Kanpur , which is one of the prestigious colleges in INDIA.
Cleared JEE Advance in 2017.I am a flexible teacher because I understand that all students learn in different ways and at different paces. When teaching, I make sure that every student has a grasp of the subject before moving on.
I will help student to get the basic understanding clear. I believe friendly behavior with student can help both the student and the teacher.
I love science and my students do the same.
4.90+
44+ Reviews
166+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
1. [10] Let A = 2 4 a b c d e f g h i 3 5, B = 2 4 d + 5g e+ 5h f+ 5i a 2g b 2h c 2i g h i 3 5, C = 2 4 2g 2h 2i 3d 3e 3f 1a 1b 1c 3 5. Suppose that det A = 5. Find det B = , det C = , and det (AC) =
-
Consider A-E, five mutually exclusive alternatives: A B C D E Initial cost $600 $600 $600 $600 $600 Uniform annual 100 100 100 150 150 Benefits for first 5 years 5 years 50 100 110 0 50 The interest...
-
it is sas and math it is like Q I FOUND here but extra Q i need help with a b c d I want to update file also but I can not but it is same what in website thanks 4. The Internet site...
-
Which of the following industries would most likely have the highest ratio of sales revenue to property, plant and equipment? Question 9 options: 1) Airline 2) Consumer product manufacturing company...
-
What is a data flow diagram (DFD) used for?
-
My employee seems to leave work anytime between 5PM and 6PM, uniformly. a) What is the probability he will still be at work at 5:45 PM? b) What is the probability he will still be at work at 5:45 PM...
-
In a vapour compression refrigeration system, the condition of refrigerant before entering the compressor is (a) saturated liquid (b) wet vapour (c) dry saturated liquid (d) superheated vapour
-
Parker, the owner of certain unimproved real estate in Chicago, employed Adams, a real estate agent, to sell the property for a price of $250,000 or more and agreed to pay Adams a commission of 6...
-
A dog training business began on December 1. The following transactions occurred during its first month. December 1 Receives $22,000 cash as an owner investment in exchange for common stock. December...
-
Rivera Company has several processing departments. Costs to be accounted for in the Assembly Department for November 2022 totaled $2,280,000 as follows. Production records show that 35,000 units were...
-
Describe some measures a firm can take to decrease its cash conversion cycle .
-
Can the cash conversion cycle be negative? Explain.
-
On August 25, Champion Company received a 90-day, 9 percent note in settlement of an account receivable in the amount of $20,000. Determine the maturity date, amount of interest on the note, and...
-
Assume the same facts as in problem 60 , except that the partnership agrees to distribute property 2 and \(\$ 75,000\) cash to Clay in liquidation of his interest. (To fund the distribution, the...
-
Explain how recruitment and selection practice can be shaped to accommodate the demands of strategic change and unplanned change arising from an uncertain future;
-
Using the ideas discussed below, how would you summarise organisational fluidity? The concept of the virtual organisation appears suited to knowledge-based work that utilises technologies such as the...
-
Evaluate evidence to determine the extent of strategic recruitment and selection practice;
-
DaimlerChryslers product portfolio ranges from small cars to sports cars and luxury sedans; and from versatile vans to trucks and coaches. Daimler- Chryslers passenger car brands include Maybach,...
-
Dori is 58 years old and retired in 2018. She receives a pension of $25,000 a year and no other income. She wishes to put the maximum allowed into an IRA. How much can she contribute to her IRA?
-
Explain five different cases of income exempt from tax with clear examples.
-
The 1 H NMR spectrum shown is that of a compound with formula C 9 H 10 O. How many double bonds and/or rings does this compound contain? If the unknown has an IR absorption at 1690 cm ?1 , what is a...
-
The 1 H NMR spectrum shown is that of a compound isomeric with the one in Problem 19.65. This isomer has an IR absorption at 1730 cm ?1 . Propose a structure. TMS 10 O ppm 1. Chemical shift (8)...
-
Propose structures for molecules that meet the following descriptions. Assume that the kinds of carbons (1, 2, 3, or 4) have been assigned by DEPT-NMR. (a) C 6 H 12 O; IR: 1715 cm 1 ; 13 C NMR: 8.0 ...
-
3. Show that : -3t = y 3 sin(t) cos(t) + e is a solution to the differential equation y' + 3 y 10 sin(t)
-
The United States, Austrailia, Canada, and the United Kingdom cluster high on individualism but low on power distance on Hofstede\'s cutlutral graph. What does this mean to someone looking to market...
-
Mini-garden project Week one of the mini-garden project involved Sacha and her fellow pre-schoolers planting sunflower seeds into the outdoor garden bed. Each child in the activity has been assigned...

Study smarter with the SolutionInn App


