Classify each of the following carbohydrates as an aldose or ketose, and then insert the appropriate term
Question:
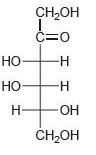
Classify each of the following carbohydrates as an aldose or ketose, and then insert the appropriate term to indicate the number of carbon atoms present (e.g., an aldopentose):
(a)
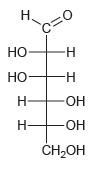
(b)
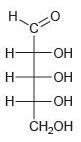
(c)
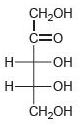
(d)
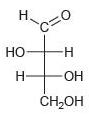
(e)
Transcribed Image Text:
H. HO -H но- H- H- OH H- -OH ČH,OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a An aldohexose ...View the full answer

Answered By

Benard Ndini Mwendwa
I am a graduate from Kenya. I managed to score one of the highest levels in my BS. I have experience in academic writing since I have been working as a freelancer in most of my time. I am willing to help other students attain better grades in their academic portfolio. Thank you.
4.90+
107+ Reviews
240+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Classify each of the following compounds as an alkane, alkene, alkyne, alcohol, aldehyde, amine, and so forth. (a) (b) CH3-C¡CH (c) (d) (e) (f) Obtained from oil of cloves -2. Sex attractant...
-
Classify each of the following as a strong acid or a weak acid. O H
-
Classify each of the following as a primary, secondary, or tertiary alcohol. a. b. c. d. CH3 CH3 CH3CCH2CHOH CH CH,OH CH,CH2CCH CH3 CH2CH3 CH3CH2COH CH2CH3
-
Remove left recursion in the following grammar. Show each step. Hint: First remove direct left recursion. Then the indirect left recursion. A AB | Aab | BA| a Bb | | b
-
Willitte Pharmaceuticals manufactures an over-the-counter allergy medication. The company sells both large commercial containers of 1,000 capsules to health care facilities and travel packs of 20...
-
Steam enters a turbine at \(700 \mathrm{kPa}, 250^{\circ} \mathrm{C}\) and exhausts at \(15 \mathrm{kPa}\). The efficiency of the turbine is 0.8. Calculate (a) the power output per \(\mathrm{kg}\) of...
-
a. Describe the circumstances where discovery sampling may be useful in auditing. b. What factors are necessary to determine sample size in discovery sampling?
-
Khonner Stores, Inc., sells electronics and appliances. The excerpts that follow are adapted from Khonner Stores financial statements for 2014 and 2013. Requirements 1. How much was Khonner Stores...
-
A steel wire with mass 29.9 g and length 1.16 m is strung on a bass so that the distance from the nut to the bridge is 1.10 m. (a) Compute the linear density of the string. kg/m (b) What velocity...
-
Lancet Engineering completed the following transactions in the month of June. a. Jenna Lancet, the owner, invested $195,000 cash, office equipment with a value of $8,200, and $80,000 of drafting...
-
Draw and name the enantiomer of d-fructose.
-
Would you expect an aldohexose and a ketohexose to be constitutionally isomeric? Explain why or why not.
-
On March 1, Sharon Fitzgerald entered into an oral lease of a house owned by Parkin. The lease was on a month-to-month basis, and the rent was set at $290 per month. Parkin also agreed to make...
-
When a cloud computing service provider receives jobs consisting of multiple Virtual Machines (VMs) (e.g., a MapReduce job), many scheduling options exist. The VMs can be scheduled in a round-robin...
-
Figure 6.12 shows the impact of user perceived response time on revenue, and motivates the need to achieve high-throughput while maintaining low latency. a. Taking Web search as an example, what are...
-
Compute the effective CPI for RISC-V using Figure A.29 and the table above. Average the instruction frequencies of perlbench and sjeng to obtain the instruction mix. Figure A.29 Program astar bzip...
-
Some applications reada large dataset first and then modify most or all of it. The base MSI coherence protocol will first fetch all of the cache blocks in the Shared state and then be forced to...
-
For this exercise, consider a simplified equation for the total operational power of a WSC as follows: a. Assume an 8 MW datacenter at 80% power usage, electricity costs of $0.10 per kilowatt-hour,...
-
Under the direct method, what did Ferrari need to show to establish a disability under the ADA? Was he successful? Why or Why not? What must Ferrari establish to succeed under the indirect method? Is...
-
Repeat Exercise 16.6 using the t-test of the coefficient of correlation. Is this result identical to the one you produced in Exercise 16.6?
-
Iodination of alkanes using iodine (I2) is usually an unfavorable reaction. Tetraiodomethane (CI4) can be used as the iodine source for iodination, in the presence of a free-radical initiator such as...
-
Under base-catalyzed conditions, two molecules of acetone can condense to form diacetone alcohol. At room temperature (25 °C), about 5% of the acetone is converted to diacetone alcohol. Determine...
-
When ethene is mixed with hydrogen in the presence of a platinum catalyst, hydrogen adds across the double bond to form ethane. At room temperature, the reaction goes to completion. Predict the signs...
-
Part 1: The Program Explain in detail: The program you identified within your specialization area(My specialization is Special Education)that is in need of evaluation. Be sure to include the purpose...
-
Should the U.S. Government play a greater role in providing an economic "safety net" for its citizens than it currently does? Alternatively, should such a greater role be the obligation of...
-
How could I Identify and describe the segmentation of the workforce in a hospital setting (drawing up our study of how to differentiate employee groups depending on their drivers, such as flexible...

Study smarter with the SolutionInn App


