Compare the structures of 1,4-pentadiene and divinyl amine: The first compound does not absorb UV light in
Question:
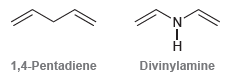
The first compound does not absorb UV light in the region between 200 and 400 nm. The second compound does absorb light above 200 nm. Using this information, identify the hybridization state of the nitrogen atom in divinylamine, and justify your answer.
'N' Divinylamine 1,4-Pentadiene
Step by Step Answer:
The nitrogen atom in divinyl amine is sp 2 hybridized The lone pai...View the full answer



Related Video
Using a few basic physics principles, you can impress your friends with a trick that makes a bottle disappear. For this experiment, you will need a mini plastic bottle, glycerin, a glass, and a funnel. First, open the bottle and pour some glycerin into it, then close the bottle tightly. Next, pour some water into the glass using a funnel. Place the mini bottle into the glass of water and it will look normal. This is because light travels through air faster than it travels through the glass and water, allowing our eyes to see the bottle inside the glass. However, when you fill the mini bottle with more glycerin, and pour glycerin into the glass, then put the glycerin filled bottle into the glass with glycerin in it. Half of the bottle that is submerged in the glycerin will become invisible as the light travels through glass and glycerin at the same speed, thus it does not bend and no refraction takes place, making the bottle invisible. This happens because both glass and glycerin have almost the same refractive index, which causes the speed of light to be the same in both mediums, causing no bending of light and making the bottle disappear.
Students also viewed these Sciences questions
-
Compare the structures of a synchondrosis and a symphysis.
-
Compare the structures of lanosterol and cholesterol, and catalog the changes needed for the transformation.
-
Compare the structures of HNO2 and H2CO3. Which would you expect to be the stronger acid? Explain your choice.
-
1. The Jarmon Company manufactures and sells a line of exclusive sportswear. Prepare a financial position as at 31st December 2022 from the following information. Account Receivable Long term loan...
-
A sample of sand has a relative density of 40% with a specific gravity of solids of 2.65. The minimum void ratio is 0.45 and the maximum void ratio is 0.97. (a) What is the unit weight (in units of...
-
The following excerpts from past equity analyst reports illustrate the use of multiple ratios in communicating views about a stocks value. In the first excerpt, from a report on Colorpak Ltd....
-
Infections Can Lower IQ A headline in June 2015 proclaims "Infections can lower IQ." The headline is based on a study in which scientists gave an IQ test to Danish men at age 19. They also analyzed...
-
On July 1, 2014, McVay Corporation issued $15 million of 10-year bonds with an 8% stated interest rate. The bonds pay interest semiannually on June 30 and December 31 of each year. The market rate of...
-
What is the most appropriate discount rate to discount cash flows at for NPV analysis?
-
The L/R time constant of the field winding of an 500-MVA synchronous generator is 4.8 s. At normal operating conditions, the field winding is known to be dissipating 1.3 MW. Calculate the...
-
How do perspectives on climate change of highly developed countries differ from those of less developed countries?
-
Provide a systematic name for each of the following compounds. a. b. c. d. e. H. CH3 Br
-
Compute the Jacobian (at the point, if indicated). (r,t) = (r sint, r- cost), (r,t)= (1,7)
-
Innovation in education may mean new and unique ideas and practices imaginations and pushes frontiers of their knowledge and understanding that expand students It means finding ways you can, being...
-
Terri and Albert plan to invest $5,000 at the end of each year in an individual retirement account earning a rate of return of 11% compounded annually. What will be the value of the account after 15...
-
What is the probability that a majority of the committee will vote to keep the president in place, if no one changes their minds?
-
Sandra receives a divorce settlement of $116,000. Her marginal tax rate is 25 percent, and the capital gains tax rate is 15 percent. How much will her account be worth in 20 years under each of the...
-
What is loan strategy that I can recommend to the client to help keep track and pay down the loans, that could also offer her a lower rate of interest?
-
Determine the due date of the loan, using the exact time, if the loan is made on the given date for the given number of days. July 4 for 150 days.
-
Gopher, Inc. developing its upcoming budgeted Costs of Quality (COQ) with the following information: Expense Item Budget Raw Materials Inspection $ 15,000 EPA Fine 200,000 Design Engineering 15,000...
-
Identify the indicated faces of carbon atoms in the following molecules as Re orSi: (b) (a) H-2 "CH2 - H Hydroxyacetone Crotyl alcohol
-
Lactic acid buildup in tired muscles results from reduction of pyruvate. If the reaction occurs from the Re face, what is the stereochemistry of theproduct? OH CH3CHCO2 H3C CO2 Lactate Pyruvate
-
The aconitase-catalyzed addition of water to cis-aconitate in the citric acid cycle occurs with the following stereochemistry. Does the addition of the OH group occur on the Re or the Si face of the...
-
If you were to enter the banking industry, you might find yourself approving or not approving loans. The following is a good example of a common event you could encounter. Company X is looking for...
-
You see a two year bond with annual coupon rate of 8% that has these flows. Years ahead Cashflows 0 ?? 0.25 0 0.5 0 0.75 1 1.25 1.5 1.75 2 8 0 0 0 108 You will be asked to calculate its price at...
-
Use synthetic division and the Remainder Theorem to evaluate P(c). P(x) = 6x + 5x +9, c = 11/1 P(1) =

Study smarter with the SolutionInn App


