Compare the wave number of absorption for the following two C = C bonds. Use resonance structures
Question:
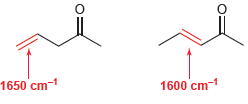
Transcribed Image Text:
1650 cm-1 1600 cm-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
The C C bond in the conjugated compoun...View the full answer

Answered By

Madhvendra Pandey
Hi! I am Madhvendra, and I am your new friend ready to help you in the field of business, accounting, and finance. I am a College graduate in B.Com, and currently pursuing a Chartered Accountancy course (i.e equivalent to CPA in the USA). I have around 3 years of experience in the field of Financial Accounts, finance and, business studies, thereby looking forward to sharing those experiences in such a way that finds suitable solutions to your query.
Thus, please feel free to contact me regarding the same.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
All of the following compounds absorb IR radiation in the range between 1600 and 1850 cm -1 . In each case, identify the specific bond(s) responsible for the absorption(s), and predict the...
-
Esters contain two CO bonds and therefore will produce two separate stretching signals in the fingerprint region of an IR spectrum. One of these signals typically appears at approximately 1000 cm -1...
-
If the wave number of the J = 1 f-- 0 rotational transition of IH81Br considered as a rigid rotator is 16.93 cm-1, what is (a) The moment of inertia of the molecule, (b) The bond length?
-
1. Insurance Act, RSBC 1996 c226 Read Parts 1 and 2 of this statute and describe any changes to the standard commonlaw rules for contracts that you notice. 2. KP Pacific Holdings Ltd. and Churchland...
-
The Sieve of Eratosthenes is a method used to compute all primes less than N. We begin by making a table of integers 2 to N. We find the smallest integer, i, that is not crossed out, print i, and...
-
Each of the statements below is true, but Dalton might have had trouble explaining some of them with his atomic theory. Give explanations for the following statements. a. The space filling models for...
-
With reference to the interrequest time data on page 29 , obtain \(95 \%\) tolerance limits on the proportion \(P=0.90\) of the population of interrequest times. Take logs, use the normal theory...
-
Assume that Nolanvilles scal year ends on December 31. 1. Nolanvilles payroll for one of its departments is $15,000 per week. It pays its employees on the Thursday of the week following that in which...
-
Authenticity involves behaving in a way that is genuine, true to one self, and that reveals one's individual character. Though the lecture focused on the benefits of authenticity, others have argued...
-
Five clowns each have a red wig and a blue wig, which they are all equally likely to wear at any particular time. Find the probability that, at any particular time: a. Exactly two clowns are wearing...
-
The type of private property that is protected by a copyright or patent is a. Land. b. Financial capital. c. Intellectual property. d. Personal property.
-
Because there are _______, adjusted gross income is always ______ taxable income. a. Deductions and exemptions; less than b. Deductions and exemptions; greater than c. Credits; greater than d....
-
For the following exercises, evaluate the function f at the indicated values f(3), f(2), f(a), f(a), f(a + h). f(x) = 2x 5
-
A Barrozo sent a tax invoice to D Anderson on 9 August for $1100 with the terms of 5% 10 days/net 30 days. A Barrozo received payment on 17 August and allowed the discount according to the terms on...
-
L Barchi sold goods to J Altimo for $550 on 15 July. A discount of $22 was to be allowed if the remittance was received by 25 July. L Barchi received the remittance from J Altimo on 25 July and...
-
On 1 February 2022, the balances of J Kelaher were: bank $4000, GST receivable $100, inventory $12 000 (the perpetual inventory system is used), plant and equipment $15 000, GST payable $200, loan...
-
P Forrester has elected to prepare and maintain the accounts using the cash basis of accounting and not on the accrual basis of accounting. Enter the following transactions, where appropriate, in the...
-
G Rettkowicz commenced business from 1 August 2022 with bank $9000, inventory $2000, motor vehicle $8000, computer $2800; process this data through the general journal and post to the general ledger....
-
1. From what you have read, how does Mr. Ma fit the profile of an entrepreneur? 2. What are the reasons for Mr. Mas success when so many others have failed (particularly in the food service...
-
Consider the function f and its graph. a. Estimate the zeros of the area function b. Estimate the points (if any) at which A has a local maximum or minimum. c. Sketch a graph of A, for 0 x 10,...
-
Starting with benzene as your only source of aromatic compounds, how would you synthesize the following substances? Assume that you can separate ortho and Para isomers if necessary. (a) p-Chloro...
-
Starting with either benzene or toluene, how would you synthesize the following substances? Assume that ortho and Para isomers can be separated. (a) 2-Brorno-4-nitrotoluene (b) 1, 3,...
-
As written, the following syntheses have flaws. What is wrong witheach? CH o (b) CI (a) .CH 1. Cl2, FeCl3 1. HNO3, H2SO4 2. CH3CI, AICI3 3. Fe, H30* 4. NaOH, H20 2. KMNO4 NH2 (c) CH3 CH NO2 1. CH3CI,...
-
Martin Company applies manufacturing overhead based on direct labor hours. Information concerning manufacturing overhead and labor for the year is as follows: Actual manufacturing overhead Estimated...
-
$ 719,000 280,000 439,000 150,400 Joyner Company's income statement for Year 2 follows: Sales Cost of goods sold Gross margin Selling and administrative expenses Net operating income Nonoperating...
-
FIFO and LIFO costs under perpetual inventory system The following units of an item were available for sale during the year: Beginning inventory 22 units at $44 Sale 18 units at $62 First purchase 18...

Study smarter with the SolutionInn App


