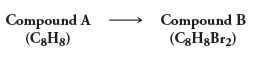
Compound A has molecular formula C 8 H 8 . When treated with excess Br 2 ,
Question:
Transcribed Image Text:
Compound A (C3Hg) Compound B (C3H&B12)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
Compound ...View the full answer

Answered By

Diane Joyce Pastorin
Please accept my enthusiastic application to solutioninn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Compound A has molecular formula C10H10O and exhibits a strong signal at 1720 cm1 in its IR spectrum. Treatment with 1,2-ethanedithiol followed by Raney nickel affords the product shown below....
-
Compound A has molecular formula C 9 H 8 O 2 and exhibits a strong signal at 1740 cm -1 in its IR spectrum. Treatment with two equivalents of LAH followed by water gives the following diol. Identify...
-
Compound A has molecular formula C 8 H 8 O. An IR spectrum of compound A exhibits a signal at 1680 cm -1 . The 1 H NMR spectrum of compound A exhibits a group of signals between 7.5 and 8 ppm (with a...
-
If I have a lump sum of $28,621 today, calculate the value of that lump sum 29 years in the future assuming earn 2.50% compounded annually on my investment?
-
A sample of soil was tested in the laboratory and the following grain size analysis results were obtained. Classify this soil according to the USCS, providing the group symbol for it.?
-
Discuss the various dimensions of transportation service and their impact upon costs for a company.
-
List the advantages and disadvantages of a jet condenser.
-
Demand for Stuffin Such stuffed animal shells and stuffing/finishing kits manufactured by RoseMarie Limited is increasing, and management requests assistance from you in determining the best sales...
-
What is the present value of a perpetual stream of cash flows that pays $6,500 at the end of year one and the annual cash flows grow at a rate of 4% per year indefinitely, if the appropriate discount...
-
There is a lottery with n coupons and n people take part in it. Each person picks exactly one coupon. Coupons are numbered consecutively from 1 to n, n being the maximum ticket number. The winner of...
-
What are the main forms of electronic communication? What are their unique benefits and challenges?
-
In some circumstances, dehydrogenation is observed. Dehydrogenation involves the loss of two hydrogen atoms (the reverse of hydrogenation). Analyze each of the following dehydrogenation reactions and...
-
If a firm uses n inputs (n > 2), what inequality does the theory of revealed cost minimization imply about changes in factor prices (w i ) and the changes in factor demands (x i ) for a given level...
-
Suppose the market portfolio is equally likely to increase by 20% or decrease by 13%. a. Calculate the beta of a firm that goes up on average by 39% when the market goes up and goes down by 29% when...
-
Your engineers are developing a new product to launch next year that will require both software and hardware innovations. The software team requests a budget of $6 million and forecasts an 80% chance...
-
You expect HGH stock to have a 20% return next year and a 45% volatility. You have $25,000 to invest, but plan to invest a total of $50,000 in HGH, raising the additional $25,000 by shorting either...
-
You own a cab company and are evaluating two options to replace your fleet. Either you can take out a five-year lease on the replacement cabs for $491 per month per cab, or you can purchase the cabs...
-
Consumer electronics is a very competitive business. What might be the success story of the year one year is a forgotten item two years later. Rapid product commoditization makes the consumer...
-
Verify that the function satisfies the hypotheses of the ean Value Theorem on the given interval. Then find all numbers c that satisfy the conclusion of the Mean Value Theorem. f(x) = x 3 - 3x + 2,...
-
How can NAFTA be beneficial to suppliers of Walmart?
-
Assign R or S configuration to each chirality center in the followingmolecules: (a) (b) ,CH (c) , . CH CHCH
-
Assign R or S configuration to each chirality center in the following biologicalmolecules: (a) (b) .C 'N' N-N - CH2CH2CH2CH2CO2 Prostaglandin E, Biotin
-
Draw tetrahedral representations of the following molecules: (a) (S)-2-Chlorohutane (b) (R)-3-Chloro-1-pentene
-
According to the video "The Importance of Budgeting," budgeting is a crucial aspect of running a successful business as it helps administrators allocate resources effectively to meet their goals. It...
-
Managing finances is essential for any business, big or small. It involves careful planning and allocation of resources to achieve goals. Budgeting is crucial as it helps firms manage their cash...
-
Your boss (for 1 of the six organizations you have to choose from-Google, Walt Disney Company, Amazon, P&G, 3M, or Ford Motor Company) has asked you to represent the organization in an industry panel...

Study smarter with the SolutionInn App


