Consider the following compound with molecular formula C 4 H 8 O: (a) Draw a constitutional isomer
Question:
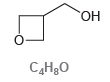
(a) Draw a constitutional isomer that you expect will be approximately one trillion (1012) times more acidic than the compound above.
(b) Draw a constitutional isomer that you expect will be less acidic than the compound above.
(c) Draw a constitutional isomer that you expect will have approximately the same pKa as the compound above.
Transcribed Image Text:
Он СДН,О
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
a b There are othe...View the full answer

Answered By

Pharashram rai
i am highly creative, resourceful and dedicated freelancer an excellent record of successful classroom presentations and writing . I have more than 4 years experience in tutoring students especially by using my note making strategies and engineering field . Especially adept at teaching methods of maths and writing , and flexible teaching style with the willingness to work beyond the call of duty. Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with .
4.80+
65+ Reviews
270+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A compound with molecular formula C4H8O has a strong IR absorption at 1730 cm-1. Its mass spectrum includes key peaks at m/z 44 (the base peak) and m/z 29. Propose a structure for the compound and...
-
A compound with molecular formula C4H6O gives the infrared spectrum shown in Figure 13.34. Identify the compound. 3.5 4 5 12 13
-
The IR spectrum of a compound with molecular formula C5H8O was obtained in CCl4 and is shown in Figure 13.42. Identify the compound. Wavelenga qum) 15 16 14 3600 340) 3800 3300 3000 280K 2600 2400...
-
If a= 3 m, determine the magnitudes of P and Q required to maintain the cable in the shape shown. DI 120 kN
-
What are the fundamental differences between brokers and dealers? Which has a higher risk of capital losses, and why?
-
Determine the steady-state response of the system governed by the following difference equation: \[12 y(n)-7 y(n-1)+y(n-2)=\sin \left(\frac{\pi}{3} n ight) u(n) \text {. }\]
-
Ronda Rousey Fight Times Perhaps the most popular fighter since the turn of the decade, Ronda Rousey is famous for defeating her opponents quickly. The five number summary for the times of her first...
-
For fiscal year 2011, Starbucks Corporation (SBUX) had total revenues of $11.70 billion, net income of $1.25 billion, total assets of $7.36 billion, and total shareholders equity of $4.38 billion. a....
-
Suppose a solenoid has a radius of 6 . 0 cm and 2 0 0 0 turns of wire per meter of length along its axis has 1 5 A passing through its wires. Determine the magnitude of the magnetic field on the...
-
According to Exhibit 4-8, which is the best chart for comparisons of earnings per share over many periods? How about for only a few periods? Exhibit 4-8 Conceptual (Qualitative) Data-Driven...
-
Consider the pK a values of the following constitutional isomers: Using the rules that we developed in this chapter (ARIO), we might have expected these two compounds to have the same pKa....
-
There are only four constitutional isomers with molecular formula C 4 H 9 NO 2 that contain a nitro group (-NO 2 ). Three of these isomers have similar pK a values, while the fourth isomer has a much...
-
Consider a simple quantum system consisting of twenty independent simple harmonic oscillators each with frequency . The energy of this system is just the sum of the energies of the 20 oscillators....
-
1-Do these resistors satisfy the Ohm's Law? Justify your answer 2-What voltage do you expect if 0.2 A goes through the 100 resistor? 3-What current do you expect if the voltage across the 200...
-
Please provide a solution to the below question. Australia is a leading country for gold mining, extracting gold from open-cut mines in the ground mainly from the state of Western Australia. The...
-
You are performing a capital budgeting analysis for a client but they are confused about two parts of your analysis. Please answer the following: a. What would constitute an opportunity cost and why...
-
What is the idea of goldensent?the visioning process Introduce the e-commerce business you have chosen. o Provide an overview of the business idea Product and type of services. What are the different...
-
A) Safeguarding your personal information: Review the PowerPoint slides in the Extra Credit Modulein Canvas. Find a current article about identity theft or safeguarding your personal information....
-
Determine the range and standard deviation of the set of data. When appropriate, round standard deviations to the nearest hundredth. 5, 9, 10, 14, 2, 11, 13, 14, 8, 4
-
Some people argue that the internal control requirements of the Sarbanes-Oxley Act (SOX) put U.S. companies at a competitive disadvantage to companies outside the United States. Discuss the...
-
The following data for isomeric four-carbon alcohols show that there is a decrease in boiling point with increasing substitution of the OH-bearing carbon. How might you account for this trend?...
-
Rank the following substances in order of increasing acidity: (a) (CH3)2CHOH, HC CH, (CF3)2CHOH, CH4OH (b) Phenol, p-methyl phenol, p-(trifluoromethyl) phenol (c) Benzyl alcohol, phenol, p-hydroxy...
-
P-Nitro benzyl alcohol is more acidic than benzyl alcohol but p-methoxy benzyl alcohol is less acidic. Explain.
-
Suppose we have two spin-1/2 particles, with associated spin operators and 2. The simultaneous eigenstates of S1 and S2z are m, m2), which we can write as It. t), It), H), H). The spins interact with...
-
Directions: Read and answer the question below. The Assignment is due on February 6, before class. Your answers should be in paragraph form and in Arial, 11 point font. Your answer should be between...
-
In Lorenzo, the Supreme Court extended liability to those who disseminate false and misleading statements with the intent to defraud, even if they are not the "maker" of the statement under the...

Study smarter with the SolutionInn App


