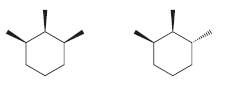
Consider the following two compounds. These compounds are stereoisomers of 1,2,3-trimethylcyclohexane. One of these compounds has three
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
The first compound has three chirality centers This is apparent if we assi...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following two compounds. When treated with NaOH, one of these compounds forms an epoxide quite rapidly, while the other forms an epoxide very slowly. Identify which compound reacts more...
-
Consider the following two compounds. How would you distinguish between them using: a) IR spectroscopy? b) 1 H NMR spectroscopy? c) 13 C NMR spectroscopy?
-
Consider the following two compounds: CH3CH2CH2CH2CH2OH 1-pentanol CH3CH2CH2CH2CH2CH3 hexane a. What are the different types of intermolecular forces that exist in each compound? b. One of these...
-
. What impacts of influences adult human behavior? Perspective Support: You must critically evaluate the issue and address the question: " What influences or impacts personality development?" by...
-
What role do finance companies play in leveraged buyouts?
-
Show that a system described by Equation (1.63) is linear if and only if the auxiliary conditions are zero. Show also that the system is time invariant if the zero auxiliary conditions are defined...
-
Which Accomplishment of LeBron James Is Most Impressive? Table 2.23 shows the means and standard deviations for four of the variables in the NBAPlayers2015 dataset. FGPct is the field goal...
-
The following information describes transactions for Morgenstern Advertising Company during July: a. On July 5, Morgenstern purchased and received $21,500 of supplies on credit from Drexel Supply...
-
One possible waste product of a nuclear reactor is the nuclide caesium-137 which decays to form a nuclide of barium (Ba). a) State the nuclear equation for this decay. b) State three quantities that...
-
Can Pepsi or any other company ever surpass Coca- Cola? Why or why not? What are Coca-Colas greatest risks?
-
Cyclopropane is a compound in which the carbon atoms form a three-membered ring: Each of the carbon atoms in cyclopropane is sp 3 hybridized. Cyclopropane is more reactive than other cyclic compounds...
-
When butyl bromide is treated with sodium iodide in ethanol, the concentration of iodide quickly decreases but then slowly returns to its original concentration. Identify the major product of the...
-
J Leeson commenced business on 15 April 2022 with $5000 in the bank, GST receivable $350 and a computer valued at $3500. 20 April: Additional funds of $15 000 were injected by J Leeson into the...
-
It costs a manufacturer $3,327 to make a product. The rate of markup is 32.00% of the selling price and it offers a markdown of 10.00% during a discount period. a. What is the regular selling price?...
-
Given that an ordinary annuity and an annuity due have the same payments and positive interest rate, it follows that the present value of the ordinary annuity is greater than the present value of an...
-
Suppose that U.S. auto producers decide to raise prices to take advantage of their comparative advantage. Given the exchange rate is P10 = $US1 and trade costs of 10%, what is the maximum price that...
-
Seymour is 76 years old. Married at the age of 23, his wife passed away five years later in a car accident, leaving Seymour alone to raise their two children. Seymour worked until he was 55 and...
-
Research and evaluate the following four insurance companies: AIG. Travelers. Saint Paul. Hartford. Determine which company you consider the most financially sound and develop a brief report...
-
Matthew purchased some items at Staples each day for five days. The mode of the number of items Matthew purchased is higher than the median of the number of items he purchased. The median of the...
-
General Electric Capital, a division of General Electric, uses long-term debt extensively. In a recent year, GE Capital issued $11 billion in long-term debt to investors, then within days filed legal...
-
What products would you expect from oxidation of the following compounds with CrO3 in aqueous acid with pyridinium chlorochromate? (a) 1-Hexanol (b) 2-Hexanol (c) Hexanol
-
TMS ethers can be removed by treatment with fluoride ion as well as be acid-catalyzed hydrolysis. Propose a mechanism for the reaction of cyclohexyl TMS ether with LiF. Fluorotrimethyisilane is a...
-
Show the mechanism of the reaction of p-methaylphenol with 2-methylpropene and H3PO4 catalyst to yield the food additive BHT.
-
Research the different processes involved during the planning phase of a project. Identify the tasks and techniques associated with each of these processes. Prepare diagram where you relate...
-
How do cultural norms and values intersect with social structure, influencing patterns of behavior, identity formation, and societal organization ?
-
We manufacture breakfast cereal at our factory in Toad Suck, AR. Our current selling price is $2.40 per box, F.O.B. our shipping dock (our customers pay shipping expenses). Our gross profit margin is...

Study smarter with the SolutionInn App


