Consider the following two compounds. When treated with NaOH, one of these compounds forms an epoxide quite
Question:
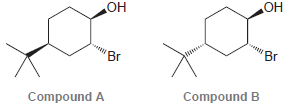
Transcribed Image Text:
OH OH 'Br 'Br Compound B Compound A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
T his process for epoxide formation involves deprotonation of the hyd...View the full answer

Answered By

Sinmon Warui Kamau
After moving up and down looking for a job, a friend introduced me to freelance writing. I started with content writing and later navigated to academic writing. I love writing because apart from making a living out of it, it is also a method of learning and helping others to learn.
5.00+
40+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following two compounds. Monochlorination of one of these compounds produces twice as many stereoisomeric products as the other. Draw the products in each case, and identify which...
-
Consider the following two compounds. These compounds are stereoisomers of 1,2,3-trimethylcyclohexane. One of these compounds has three chirality centers, while the other compound has only two...
-
Consider the following two compounds. How would you distinguish between them using: a) IR spectroscopy? b) 1 H NMR spectroscopy? c) 13 C NMR spectroscopy?
-
Determine whether the statement is true or false. If it is true, explain why. If it is false, explain why or give an example that disproves the statement.
-
PepsiCo ran an ad and promotional campaign in 1996 called "Drink Pepsi Get Stuff" campaign. The enormously successful campaign allowed customers to claim prizes in exchange for points on PepsiCo...
-
Baxtell Company manufactures and sells a single product. The following costs were incurred during the companys first year of operations: During the year, the company produced 25,000 units and sold...
-
In Example 12.3, how will the maximum height, \(z_{1}\), that the pump can be located above the water surface change if the water temperature is decreased to \(40^{\circ} \mathrm{F}\)? Example 12.3 A...
-
Stan Company has accumulated the following budget data for the year 2012. 1. Sales: 30,000 units, unit selling price $80. 2. Cost of one unit of finished goods: Direct materials 2 pounds at $5 per...
-
The pretrial hearings were set. The hearing was pertaining to both cases against me which consisted of seven counts. My dad also stood accused on the Indiana indictment which consisted of just 4 of...
-
Describe the design of a controlled experiment to determine whether the use of vitamin D supplements reduces the chance of broken bones in women with osteoporosis (weak bones). Assume you have 200...
-
Assume that the equation of state for a gas can be written in the form P(V m b(T)) = RT. Derive an expression for = 1/V (V /T)P and = 1/V (V /P)T for such a gas in terms of b(T), db(T)/dT, P, and...
-
One mole of Ar initially at 310. K undergoes an adiabatic expansion against a pressure P external = 0 from a volume of 8.5 L to a volume of 82.0 L. Calculate the final temperature using the ideal gas...
-
One Day Laundry Services purchased a new steam press on January 1, for $42,000. It is expected to have a five-year useful life and a $2,000 salvage value . One Day expects to use the steam press more...
-
Individual/class roundtable discussion: It has been said that information is power and that the separation between the economically well-off nations, individuals, companies, and the like, and those...
-
You are a systems analyst in a software development company that has been hired to do the requirements analysis phase for a large organization. What are three categories of existing documentation...
-
Surveys and questionnaires are frequently used to gather facts. What are some of the advantages and disadvantages of questionnaires? When might you choose free-format questionnaires over fixedformat...
-
What are some of the reasons to use joint requirements planning (JRP) as a fact-finding technique? What should be the basis for selecting which users and managers will participate in the JRP session,...
-
Give examples of activities covered by ESG and sustainability that are not climate risk issues.
-
What is the largest possible result of multiplying two unsigned N-bit numbers?
-
Suppose a population of bacteria doubles every hour, but that 1.0 x 106 individuals are removed before reproduction to be converted into valuable biological by-products. Suppose the population begins...
-
Poly(ethylene terephthalate), or PET, is a polyester used to make soft-drink bottles. It is prepared by reaction of ethylene glycol with 1,4-benzenedicarboxylic acid (terephthalic acid). Draw the...
-
Show the mechanism of the nucleophilic addition reaction of an alcohol with an iso-cyanate to yield a urethane.
-
What product would you expect to obtain from catalytic hydrogenation of natural rubber? Would the product be syndiotactic, atactic, or isotactic?
-
Joey set up a lawn-mowing business in his neighborhood. He currently has 7 customers that want their lawns mowed each week, for which he charges $25.00 each. Joey spends $10.00 a week in gas and...
-
Vanessa has an outstanding bond with a 7.10 % semi-annual coupon and has 16 years left to mature. The current market rate is 10.50% per annum. What is the current yield of Vanessa's Bond.
-
Estimate the hydrogen consumption required to completely remove the sulfur from a hydrotreater feedstock and to reduce the nitrogen content of the product to 15 ppm by weight. The 48.5 API naphtha...

Study smarter with the SolutionInn App


