Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the
Question:
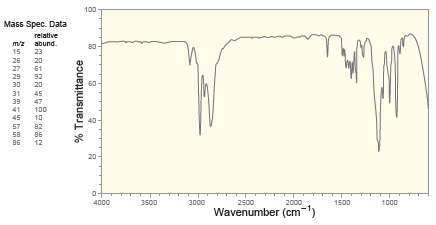
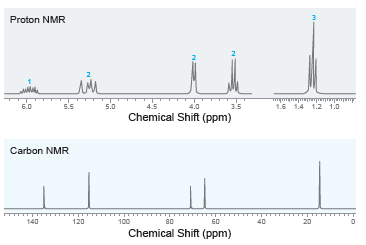
Transcribed Image Text:
100 Mass Spec. Data relative mz abund. 15 23 26 27 29 20 61 92 30 20 31 39 47 41 100 45 10 57 82 58 86 86 12 20 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm-") % Transmittance Proton NMR 1.5 14 1.2 1.0 6.0 5.5 5.0 4.5 4.0 3.5 Chemical Shift (ppm) Carbon NMR 140 120 100 80 60 20 Chemical Shift (ppm)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (18 reviews)

Answered By

Dinesh F
I have over 3 years of professional experience as an assignment tutor, and 1 year as a tutor trainee.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Deduce the structure of a compound with molecular formula C 9 H 10 O 2 that produces the following 1 H NMR spectrum and 13 C NMR spectrum: Proton NMR 10 Chemical Shift (ppm) Carbon NMR - 128.4 128.8-...
-
The IR spectrum of a compound with molecular formula C5H8O was obtained in CCl4 and is shown in Figure 13.42. Identify the compound. Wavelenga qum) 15 16 14 3600 340) 3800 3300 3000 280K 2600 2400...
-
The 1 H NMR spectrum of a compound with molecular formula C 7 H 15 C l exhibits two signals with relative integration 2 : 3. Propose a structure for this compound.
-
You work for North Carolina Parks and Recreation. Do you support or oppose the idea to unionize federal and state employees? Explain your thoughts below.
-
What are two types of information available on the Web? Which type can be censored?
-
How has trade between the United States and the European Union developed? The file EU Imports contains U.S. imports ($ M) from the European Union countries from 1997 to 2012. (Source: Download from...
-
Note that to first \(\operatorname{order} N(d)=\frac{1}{2}+d / \sqrt{2 \pi}\). Use this to derive the value of a call option when the stock price is at the present value of the strike price; that is,...
-
Imax Corporation is a large entertainment technology company, with headquarters in New York and Toronto, and theatres worldwide. Its share price, which was as high as Can.$ 13.89 on the Toronto Stock...
-
What are the two most interesting guidelines that relate to a strategic planning team formulating a vision of success? Why did you find these two interesting? What are the desired outcomes of an...
-
Walgreen Company is a well-known drugstore chain. A condensed balance sheet for August 31, 2011, follows ($ in millions): Use a format similar to Exhibit to analyze the following transactions for the...
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) Discuss. OMe . CI H,
-
Consider the structure of N,N-dimethylformamide (DMF): We might expect the two methyl groups to be equivalent; however, both the proton and carbon NMR spectra of DMF show two separate signals for the...
-
How do expansionary, tight, contractionary, and loose monetary policy affect aggregate demand?
-
You expect KStreet Cos trade at $98 per share right after paying a $2.25 dividend per share in one year. What is the most you would pay to buy the stock now if you want to earn at least a 12% return?
-
Your portfolio consists of 120 shares of CSH and 60 shares of EJH, which you just bought at $21 and $29 per share, respectively. a. What fraction of your portfolio is invested in CSH? In EJH? b. If...
-
HNL has an expected return of 17% and KOA has an expected return of 23%. If you create a portfolio that is 65% HNL and 35% KOA, what is the expected return of the portfolio?
-
Suppose that a $150,000 expenditure today on insulating layers to prevent toxic landfill seepage would eliminate $1 million worth of healthcare costs 20 years from now. If the social discount rate...
-
Fremont Enterprises has an expected return of 16% and Laurelhurst News has an expected return of 19%. If you put 48% of your portfolio in Laurelhurst and 52% in Fremont, what is the expected return...
-
Generally, the tax rate that applies to the unearned income of a minor under the kiddie tax in 2018 is: a. The same as the parents tax rate. b. The same as the single taxpayer rate. c. The same as...
-
Open Text Corporation provides a suite of business information software products. Exhibit 10-9 contains Note 10 from the companys 2013 annual report detailing long-term debt. Required: a. Open Text...
-
The heat of hydrogenation for allene (Problem 6.37) to yield propane is 295kJ/mol, and the heat of hydrogenation for a typical monosubstituted alkene such as propene is 126kJ/mol. Is allene more...
-
Predict the major product in each of the following reactions: CH (a) H20 CHCH-CH3H2CH H2SO4 (Addition of H20 occurs.) (b) CH-CH CH (c) HBr (d) 2 HCI %3 CHCH2CH2CH2CH%3DCH2
-
Predict the major product from addition of HBr to each of the following alkenes: (a) CH2 (b) (c) CH CH3CHHHH
-
Privack Corporation has a standard cost system in which it applies overhead to products based on the standard direct labor-hours allowed for the actual output of the period. Data concerning the most...
-
HomeLife Life Insurance Company has two service departments ( actuarial and premium rating ) and two production departments ( advertising and sales ) . The distribution of each service department s...
-
Otsego Industries manufactured 300,000 units of product last year and identified the following costs associated with the manufacturing activity: Variable costs: Direct materials used Direct labor...

Study smarter with the SolutionInn App


