For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic,
Question:
(a)
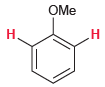
(b)
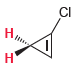
(c)
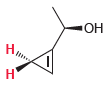
(d)
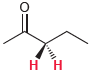
(e)
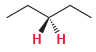
Discuss.
Transcribed Image Text:
OMe Н. Н CI H,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a Homotopic b ...View the full answer

Answered By

Ankur Gupta
I have a degree in finance from a well-renowned university and I have been working in the financial industry for over 10 years now. I have a lot of experience in financial management, and I have been teaching financial management courses at the university level for the past 5 years. I am extremely passionate about helping students learn and understand financial management, and I firmly believe that I have the necessary skills and knowledge to effectively tutor students in this subject.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following compounds, draw the important resonance forms. Indicate which structures are major and minor contributors or whether they have the same energy. (a) H2CNN (b) H2C=CH-NO2 (c)...
-
For each of the following compounds, give the systematic name and the common name (for those that have common names), and indicate whether the amines are primary, secondary, or tertiary: a. b. c....
-
For each of the following compounds, use the nitrogen rule to determine whether the molecular weight should be even or odd. Then calculate the expected m/z value for the molecular ion. a. b. c. d. O:...
-
Engineering is a dynamic field that requires continuous learning. Discuss how you plan to acquire and apply new knowledge as needed throughout your engineering career. Address the strategies you...
-
What is intellectual property? What are three examples of intellectual properties?
-
A time series plot of monthly crude oil price ($/ barrel) from January 2001 to March 2007 is shown here. Using these data, a) Fit a first-order autoregressive model. b) Obtain a forecast for March...
-
Gavin Jones is inquisitive and determined to learn both the theory and the application of investment theory. He pressed the tree farmer for additional information and learned that it was possible to...
-
State the effect (cash receipt or payment and amount) of each of the following transactions, considered individually, on cash flows: a. Sold equipment with a book value of $78,000 for $94,000. b....
-
What do you think you can do in your leadership role to empower others? What is your definition of a servant leader? What are your personal leadership strengths? What roles do you allow fellow staff...
-
A mixture of chloroform (CHC13) and acetic acid at 18oC and 1 atm (101.3 kPa) is to be extracted with water to recover the acid.(a) Forty-five kilograms of a mixture containing 35 wt% CHC13 and 65...
-
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. Data from the mass spectrum are also provided. 100 Mass Spec. Data...
-
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. Data from the mass spectrum are also provided. 100 Mass Spec. Data...
-
A 50-lb sphere A of radius 4.5 in. moving with a velocity of magnitude v0 = 6 ft/s strikes a 4.6-lb sphere B of radius 2 in. which is hanging from an in extensible cord and is initially at rest....
-
Assume that Cola Co. has a share price of $42.64. The firm will pay a dividend of $1.21 in one year, and you expect Cola Co. to raise this dividend by approximately 6.4% per year in perpetuity. a. If...
-
Suppose the dollar exchange rates of the euro and the yen are equally variable. The euro, however, tends to depreciate unexpectedly against the dollar when the return on the rest of your wealth is...
-
Summit Systems has an equity cost of capital of 11%, will pay a dividend of $1.75 in one year and its dividends had been expected to grow by 6.5% per year. You read in the paper that Summit has...
-
Apnex, Inc., is a biotechnology firm that is about to announce the results of its clinical trials of a potential new cancer drug. If the trials were successful, Apnex stock will be worth $74 per...
-
You have just purchased a share of stock for $19.05. The company is expected to pay a dividend of $0.63 per share in exactly one year. If you want to earn a 9.2% return on your investment, what price...
-
Which of the following is not a tax preference or adjustment item for the individual alternative minimum tax computation? a. Depreciation of certain assets. b. State income taxes. c. State income tax...
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
Normally, a Trans alkene is more stable than its cis isomer Trans-Cyclooctene, however, is less stable than cis-Cyclooctene by 38.5kJ/mol. Explain.
-
Trans-Cyclooctene is less stable than cis-Cyclooctene by 38.5kJ/mol, but Trans cyclononene is less stable than cis-cyclononene by only 12.2kJ/mol. Explain.
-
Allene (1, 2-propadiene), H2C = C = CH2, has two adjacent double bonds. What kind of hybridization must the central carbon have? Sketch the bonding orbitals in allene. What shape do you predict for...
-
The Kuwait Finance House (KFH) had the following transactions during the month of July 2015: 1) A customer withdraws KD 4,000 in cash from his saving account. 2) A customer deposited KD 7,000 to his...
-
4) Soundproofing in a studio reduces ambient noise from 41 dB to 30 dB. What percent of the ambient sound intensity is still making it through? Round your answer to the nearest 1%.
-
Following are the financial data for Binari Jaya Sdn Bhd as at 3 1 December 2 0 2 2 . Revenue (Sales) RM 1, 370, 100 Account Receivable RM 540, 000 Long Term Loan RM 690, 000 Cost of goods sold RM...

Study smarter with the SolutionInn App


