Deduce the structure of a compound with molecular formula C 8 H 10 O that exhibits the
Question:
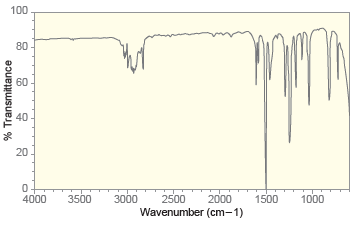
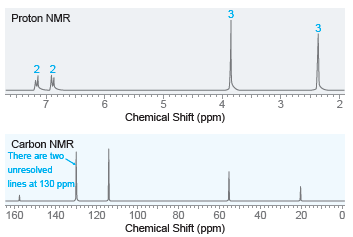
Transcribed Image Text:
100 80 60 40 20 2500 2000 Wavenumber (cm-1) 1000 4000 3500 3000 1500 % Transmittance 3 Proton NMR 3 2 2 Chemical Shift (ppm) Carbon NMR There are two, unresolved lines at 130 ppm 160 140 120 100 80 60 40 20 Chemical Shift (ppm)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)

Answered By

Asd fgh
sadasmdna,smdna,smdna,msdn,masdn,masnd,masnd,m asd.as,dmas,dma.,sd as.dmas.,dma.,s ma.,sdm.,as mda.,smd.,asmd.,asmd.,asmd.,asm
5.00+
1+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Deduce the structure of a compound with molecular formula C 6 H 14 O 2 that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 3000 2500 Wavenumber (cm-1) 4000 3500 2000 1500...
-
Deduce the structure of a compound with molecular formula C 6 H 14 O 2 that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 4000 3500 3000 2500 2000 1500 1000 Wavenumber...
-
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. Data from the mass spectrum are also provided. 100 Mass Spec. Data...
-
public class Class extends ClassA public classB ( ( Time left 0 0 : 1 2 : 0 3 int init = 1 0 ; super ( 4 0 ) ; O a . . The method super is not defined. b . . No values may be passed to super. O c . ....
-
What are three advantages of telecommuting?
-
BigCo is planning an acquisition of the assets of SmallCo and plans to hire SmallCos management team. Which of the following areas of the law may have an impact on its acquisition planning? a. Tax...
-
Fill in the Blank. Cork is a(n) ____________ isolator.
-
D&R Corp. has annual revenues of $275,000, an average contribution margin ratio of 34%, and fixed expenses of $100,000. Required: a. Management is considering adding a new product to the company's...
-
Flight Caf Planning Budget For the Month Ended July 31 27,000 $ 108,000 Budgeted meals (q) Revenue ($4.00q) Expenses: Raw materials ($1.80q) Wages and salaries ($6,100 + $0.20q) Utilities ($2,000 +...
-
Order the following functions by growth rate: N, N, N1.5, N2, N logN, N log logN, N log2 N, N log(N2), 2/N, 2N, 2N/2, 37, N2 logN, N3. Indicate which functions grow at the same rate.
-
Look back at Figure 2, which shows the inverse relationship between ticket prices and game attendance at Gigantic State University. (a) Interpret the meaning of both the slope and the intercept. (b)...
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) Discuss. OMe . CI H,
-
Solve each differential equation by variation of parameters. 3y'' - 6y' + 6y = e x sec x
-
Let f(x)=2(5)-1. Evaluate f(3) without using a calculator. Provide your answer below: f(3) = I
-
Solve the equation: 10m -5-1=2
-
Solve for v. 2 7 3 5 V 9 4
-
A survey asked 410 students whether they would eat food served in the school cafeteria. The results are recorded in the table shown. Grade Yes No 9th 67 33 10th 63 47 11th 32 68 12th 12 88 What is...
-
What is the factor form of f ( x ) = 1 6 x 1 1 - 8 1 n
-
Evaluate the expression for p = -4, q = 8, and r = -10. -p 2 - 2q + r
-
Use critical values to test the null hypothesis H0: 1 2 = 20 versus the alternative hypothesis H0: 1 2 20 by setting a equal to .10, .05, .01, and .001. How much evidence is there that the...
-
Draw structures corresponding to the following systematic names: (a) (4E)-2, 4-Dimethyl- 1, 4-hexadiene (b) cis-3, 3-Dimethyl-4-propyl-1, 5-octadicne (c) 4-Methyl-1, 2-pentadiene (d) (3E, 5Z)-2,...
-
Menthene, a hydrocarbon found in mint plants, has the systematic name 1-isopropyl-4-methylcyclohexene. Draw its structure.
-
Draw and name the 6 pentene isomers, C5H10, including E, Z isomers.
-
Discuss the challenges and opportunities of scaling up process intensification concepts from laboratory-scale experiments to industrial-scale applications, addressing issues related to...
-
Suppose the pressure in the esophagus is - 1.95 mm Hg while that in the stomach is + 18.5 mm Hg. To what height, in centimeters, could stomach fluid rise in the esophagus, assuming it has a density...
-
When light of wavelength190nm falls on acesiumsurface, electrons having a maximum kinetic energy of 4.39eV are emitted. Find values for the following. (a) the work function of cesium eV? (b) the...

Study smarter with the SolutionInn App


