Draw a mechanism for the last step of the Gabriel synthesis, performed under basic conditions. .cooe NaOH
Question:
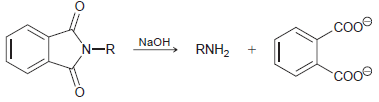
Transcribed Image Text:
.cooe NaOH RNH, N-R cooe
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
HN H D H O...View the full answer

Answered By

Rahul Sharma
I been teaching as a teacher before and had five year of experience in social sciences, now moved to college level teaching and teaching geography and other humanities subjects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a mechanism for the following reaction:
-
Give the mechanism for the last step in the biosynthesis of isopentenyl pyrophosphate, showing why ATP is required.
-
Draw a mechanism for the acidic hydrolysis of the magnesium salt shown above to acetophenone.
-
Consider a random walk consisting of equi-probable p = q = 1/2 steps in left or right directions. However the step length at ith step is given by e-, i = 1,2,3..... N, with > 0 a constant. Calculate...
-
1. All of the landline telephone numbers in a small town use the same three-digit prefix. How many different telephone numbers are possible by changing only the last four digits? 2. A college student...
-
The owner of an ice cream store asks 75 people which flavor of ice cream they prefer. Thirteen of them say strawberry, 11 say chocolate, 24 say vanilla, and 27 provide a flavor other than strawberry,...
-
Determine the vertical displacement of joint \(C\). The truss is made from A-36 steel bars having a cross-sectional area of \(150 \mathrm{~mm}^{2}\). Af H F 2 m B -1.5 m 1.5m C 1.5 m-1.5 m 6 kN 6 kN...
-
The April transactions of Wiset Company are described in Problem 7- 1A. In Problem 7- 1, Wiset Company completes these transactions during April of the current year (the terms of all its credit sales...
-
As the new sheriff, you must decide your department's policy on a range of controversial issues. Many of these issues are handled differently from one department to the next. For this discussion,...
-
d) A gas flows at the rate of 15 kmol/s at 298 K and 1 atm with a H,S content of 0.10 mol%. 95% of the H;S is to be removed by absorption with a pure liquid at 298 K. The design liquid flow rate will...
-
Propose a synthesis for each of the following transformations: (a) (b) N. -
-
One variation of the Gabriel synthesis employs hydrazine to free the amine in the final step of the synthesis. Draw the by-product obtained in this process. ? RNH, + N-R H,N-NH2
-
Calculate the Thevenin equivalent impedance ZTh in the circuit shown in figure. -j3 n 3S
-
What causes the company's finished goods inventory balance to decrease? The balance decreases when new product lines are added to the inventory mix The balance decreases when payroll is paid The...
-
Mauro Products distributes a single product, a scarf; its selling price is $16 and its variable cost is $11 per unit. The company's monthly fixed expense is $4,700. Required: 1. Solve for the...
-
Stone Beauty, Inc. is a merchandiser of stone ornaments. The company sold 8,000 units during the year. The company has provided the following information: Sales Revenue Purchases (excluding freight...
-
Super Sales Company is the exclusive distributor for a high-quality knapsack. The product sells for $120 per unit and has a CM ratio of 25%. The company's fixed expenses are $405,000 per year. The...
-
What are the security implications of context switching, particularly in terms of isolating process states and preventing unauthorized access to sensitive information during the context switch...
-
Let f(x) = 3x - x 2 and let R be the region bounded by the graph of f and the x-axis on the interval [0, 3]. a. Find the area of the surface generated when the graph of f on [0, 3] is revolved about...
-
Define a traverse in Surveying?
-
Draw complete Lewis structures for the following condensed structural formulas. (a) CH3(CH2)3CH(CH3)2 (b) (CH3)2CHCH2CI (c) CH3CH2CH(CH3)2 (d) CH2CHCHO (e) (CH3)3CCOCHCH2 (f) CH3COCOOH (g) 1CH3CH2...
-
(a) Use your molecular models to make ethane, and compare the model with the preceding structures. (b) Make a model of propane (C3H8), and draw this model using dashed lines and wedges to represent...
-
Two isomers of 1,2-dichloroethene are known. One has a dipole moment of 2.4 D; the other has zero dipole moment. Draw the two isomers and explain why one has zero dipole moment. CHCl=CHCl 1,...
-
On January 1, 2023, Bertrand, Incorporated, paid $86,900 for a 40 percent interest in Chestnut Corporation's common stock. This investee had assets with a book value of $229,500 and liabilities of...
-
Landen Corporation uses a job-order costing system. At the beginning of the year, the company made the following estimates: Direct labor-hours required to support estimated production Machine-hours...
-
Explain the concept of gene-environment interactions, and how these interactions influence the expression of complex traits and the risk of developing multifactorial diseases ?

Study smarter with the SolutionInn App


