Draw a plausible mechanism for each of the following transformations: (a) (b) (c) HJo+ N' H.
Question:
(a)
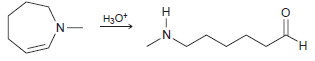
(b)
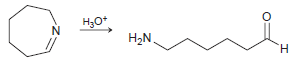
(c)
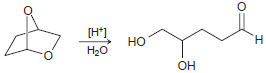
Transcribed Image Text:
HJo+ N' H. エ エーZ Но H2N. H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
a b c H 0 ...View the full answer

Answered By

Parvatht Sreekumar
I am a postgraduate in Physics with high marks. I have around 3 years of experience in teaching.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a plausible mechanism for each of the following transformations: a. b. c. d. e. Pyridine CI
-
Propose a plausible mechanism for each of the following reactions: a. b. Br Br2 . [H,SO,]
-
Propose a plausible mechanism for each of the following transformations. a. b. c. d. e. f. 1) EtMgBr 2) - 1) NaH OEt 2) EtI
-
Perform the indicated operations and then simplify. Assume that all variables represent nonzero real numbers. 20yx3 + 15y4x + 25yx4 10yx
-
Analyze and evaluate each case independently by providing the following: Facts of the case Issues Rule Analyze and explain the challenges with freedom of speech. Analyze and explain any challenges...
-
What conflicts can arise between using DCF methods for capital budgeting decisions and accrual accounting for performance evaluation? How can these conflicts be reduced?
-
Data relative to three sampling plans are presented below: Required: Calculate sample size in each of the plans. Show computations. Materiality Size of population Desired reliability Estimated...
-
You are provided with the following information for Pavey Inc. for the month ended October 31, 2010. Pavey uses a periodic method for inventory. Instructions(a) Calculate (i) ending inventory , (ii)...
-
If you receive $585 each 6 months for 1 year and the discount rate is 0.09%, what is the present value? If you receive $236 each month for 12 months and the discount rate is 4.7%, what is the present...
-
Project Alpha has two phases. You may invest in the first, in both, or in neither. The first phase requires an investment of $100 today. One year later, Alpha will deliver either $120 or $80, with...
-
Identify all of the products formed when the compound below is treated with aqueous acid: N- excess H,O+
-
Predict the major product(s) for each of the following reactions: (a) (b) (c) (d) (e) (f) CH NH2 [H*] (-H20) 1) PhMgBr 2) H20
-
Mountain Biking May Reduce Fertility in Men, Study Says was the headline of an article appearing in the San Luis Obispo Tribune (December 3, 2002). This conclusion was based on an Austrian study that...
-
Alice wants to keep the inside of her apartment at a nice temperature of 72.6F while the outside temperature is 87.3 F. If she were to get the internal temperature to 72.6 F and then turn off her air...
-
A retail company wants to determine if the average daily sales of their new product line have significantly increased compared to the old product line. The company randomly selects 100 stores that...
-
Which of the following statements regarding investment risk is correct? 1.) Beta is a measure of systematic, non-diversifiable risk 2.) Rational investors will form portfolios and eliminate...
-
For the year ended 31 December 2020 HKU Company had: Net income for the year of $18,120,000 8,705,000 ordinary shares outstanding at the beginning of 2020 450,000 outstanding cumulative and...
-
On January 1, 2024, the Excel Delivery Company purchased a delivery van for $33,000. At the end of its five-year service life, it is estimated that the van will be worth $3,000. During the five-year...
-
Sketch the vector field F by drawing a diagram like Figure 5 or Figure 9. F(x, y) = 2/1 - i + (y - x) j
-
Parkin Industries, a U.S. company, acquired a wholly-owned subsidiary, located in Italy, at the beginning of the current year, for 200,000. The subsidiary's functional currency is the euro. The...
-
(a) What two diastereomeric products could be formed in the hydroboration-oxidation of the following alkene? (b) Considering the effect of the methyl group on the approach of the borane-THF reagent...
-
The ÎG° for the equilibrium in Fig. P7.67ais 4.73 kJ mol-1 (t.13 kcal mol-1. (The equilibrium favors conformation A.) (a) Which behaves as if it is larger, methyl or phenyl (Ph)? iWhy is...
-
For the compounds in Problem 7 .6, draw a boat conformation. Problem 7.6 Cis- I,3-dimethylcyclohexane
-
Find the complete solution of the linear system, or show that it is inconsistent. (If the system has infinitely many solutions, express your answer in terms of t, where x = x(t), y = y(t), and z=t....
-
1. What is the difference between invitation to treat and offer? 2. Explain the statement "acceptance must be absolute and unqualified." Question 3 Ali goes to the convenience store to buy a can of...
-
The firm sold one of its fixed assets just as the year ended. Would this imply that operating cash flow for the year was a larger figure, a smaller figure, or would have no effect on its operating...

Study smarter with the SolutionInn App


