Predict the major product(s) for each of the following reactions: (a) (b) (c) (d) (e) (f) CH
Question:
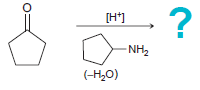
(a)
![CHз NH2 [H*] (-H20)](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1524/7/1/8/6025ae15c0a95d891524718586667.jpg)
(b)
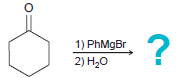
(c)
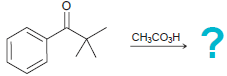
(d)
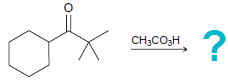
(e)
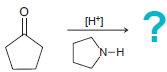
(f)
Transcribed Image Text:
CHз NH2 [H*] (-H20) 1) PhMgBr 2) H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
a b c ...View the full answer

Answered By

Joemar Canciller
I teach mathematics to students because I love to share what I have in this field.
I also want to see the students to love math and be fearless in this field.
I've been tutoring these past 2 years and I would like to continue what I've been doing.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH NH2, cat. HA N-H, cat. HA NH2 cat. HA PPha (1) HS SH (2) Raney Ni, H2 0 CH2PPha (excess) O. O...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) HO OH NH2 HA (cat.) (1) HCN (2) LiAIH4 (3) H2o mCPBA
-
Predict the major product for each proposed Diels-Alder reaction. Include stereochemistry where appropriate. (a) (b) (c) Ph 0 Ph
-
For each polynomial function, find (a) (-1), (b) (2), and (c) (0). f(x) = x + 5x
-
1. It has been said that the telephone system was analogous to a railroad and the Internet is analogous to the interstate highway system. Identify an attribute of the telephone system that is similar...
-
What are the payback and discounted payback methods? What are their main weaknesses?
-
Data pertaining to four populations are presented below: Required: For each population, a. Compute the new precision \(\left(A^{\prime \prime} ight)\) and the precision interval. b. Indicate whether...
-
Refer to the information given in the preceding case for Huron Chalk Company. In the preceding case Required: 1. Reconcile Hurons operating income reported under absorption and variable costing,...
-
The Miller Family Budget 20%5%20%35%20%Food20%Entertainment5%Utilities20%Savings20%Housing35% Data Percentage Food 0.2 Entertainment 0.05 Utilities 0.2 Savings 0.2 Housing 0.35 If the Milller's spend...
-
BusinessWeek conducted a survey of graduates from 30 top MBA programs (BusinessWeek, September 22, 2003). On the basis of the survey, assume that the mean annual salary for male and female graduates...
-
Draw a plausible mechanism for each of the following transformations: (a) (b) (c) HJo+ N' H. Z H2N. H.
-
Identify the starting materials needed to make each of the following acetals: (a) (b) (c) OEt
-
According to the liquidity premium theory, if the yield on both one-and two-year bonds are the same, would you expect the one-year yield in one-years time to be higher, lower or the same? Explain...
-
Assume that the City of Coyote has produced its financial statements for December 31, 2024, and the year then ended. The city's general fund was only used to monitor education and parks. Its capital...
-
Tamarisk Corporation had the following activities in 2020. 1 Sale of land $195,000 4. Purchase of equipment $446,000 2 Purchase of inventory $760,000 5. Issuance of common stock $302,000 3. Purchase...
-
Doorman Sportswear's articles of incorporation authorize the company to issue 6,000 $7 preferred shares and 850,000 common shares. Doorman issued 1,200 preferred shares at $100 per share. It issued...
-
A manufactured product has the following information for June. Direct materials Direct labor Overhead Units manufactured Standard Quantity and Cost 6 pounds @ $8 per pound 2 DLH @ $16 per DLH 2 DLH @...
-
The following information pertains to questions 30 & 31 Support Depts Human Resources Information Systems Budgeted Costs $72,700 $234,400 Corporate Sales $998,270 Operating Depts Consumer Sales...
-
Sketch the vector field F by drawing a diagram like Figure 5 or Figure 9. F(x, y) = 1/2x i + y j
-
5. Convert the following ERD to a relational model. SEATING RTABLE Seating ID Nbr of Guests Start TimeDate End TimeDate RTable Nbr RTable Nbr of Seats RTable Rating Uses EMPLOYEE Employee ID Emp...
-
Calculate the energy difference between cis - 1,4 - dimethylcyclohexane and the more stable conformation of trans - 1,4 - dimethylcyclohexane. .
-
The boiling poins of the l,2-dichloroethylene stereoisomers are47.4oCand 60.3C. Give the structure of the stereoisomer with the higher boiling point. Explain.
-
Label each of the following molecules as a hydrogen-bond acceptor, donor, or both. Tndicate the hydrogen that is donated or the atom that serves as the hydrogen-bond acceptor. (a) (b) (c) H3c -...
-
Stanford would like to retire in 3 5 years. He recently started a new and exciting job as a marketing specialist at an international company based in Cape Town and would like to start a retirement...
-
After many years, Unilever displays the image of a company that works for good causes. Unilever's ideals are very simple, but have a clear vision, namely promoting sustainable living. Unilever...
-
What are the roles that can be taken by intermediaries in supply chain management? How does supply chain management help to create value? How are the concepts of push and pull different when it comes...

Study smarter with the SolutionInn App


