Draw all lone pairs on each of the oxygen atoms in the compounds below. Before doing this,
Question:
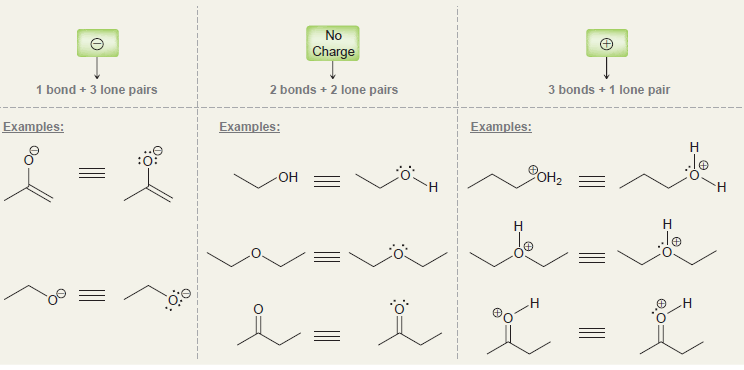
a.
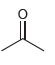
b.
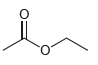
c.
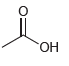
d.
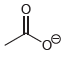
e.
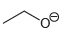
f.
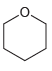
g.
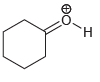
h.
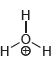
i.
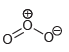
j.
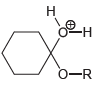
Transcribed Image Text:
No Charge 3 bonds + 1 lone pair 1 bond + 3 lone pairs 2 bonds + 2 lone pairs Examples: Examples: Examples: Н eона Он н Н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a b ...View the full answer

Answered By

ALBANUS MUTUKU
If you are looking for exceptional academic and non-academic work feel free to consider my expertise and you will not regret. I have enough experience working in the freelancing industry hence the unmistakable quality service delivery
4.70+
178+ Reviews
335+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw all lone pairs on each of the nitrogen atoms in the compounds below. First, review in the following table, and then come back to these problems. Try to identify all lone pairs without having to...
-
Sulfur dioxide (SO2) consists of two oxygen atoms (each of mass 16 u, where u is defined in Problem 39) and a single sulfur atom (of mass 32 u). The center to-center distance between the sulfur atom...
-
Each of the following compounds contains both oxygen and nitrogen atoms. Identify all lone pairs in each of the following compounds: a. b. c. d. e. f. N. O=C=N
-
At January 1, 2024, Mahmoud Industries, Inc., owed Second BancCorp $12 million under a 10% note due December 31, 2026. Interest was paid last on December 31, 2022. Mahmoud was experiencing severe...
-
On January 1, 20X1, Big Company (Big) bought 30% of the outstanding stock of Little Company (Little) for $110,000 which provided Big with the ability to significantly influence the decisions of...
-
You and a friend are asked to draw the two-dimensional electric field line partern for two charged objects located near each other. The charge on the first object is \(+2 q\) (with \(q\) positive),...
-
Assuming nominal annual yields of \(700,000,5,500,000\), and \(3,800,000 \mathrm{~kg} / \mathrm{km}^{2}\) for corn grain, sugar cane, and sweet sorghum respectively, which conversion pathway is the...
-
A paper recycling company converts newspaper, mixed paper, white office paper, and cardboard into pulp for newsprint, packaging paper, and print-stock quality paper. The recycler is currently trying...
-
Through this assignment, we will explore trends concerning an individually selected area of policy and provide context on the public policy theories at play. You will answer the following question:...
-
A company has decided to order 360 units whenever the on-hand inventory falls to 90 units. There appears to be no seasonal fluctuation to the demand, but it does fluctuate daily and is approximately...
-
For each of the compounds below determine whether any of the oxygen atoms bear a formal charge: a. b. c. d. ::
-
A carbene is a highly reactive intermediate in which a carbon atom bears a lone pair and no formal charge: How many hydrogen atoms are attached to the central carbon atom above?
-
A student builds a toy geyser for her engineering class that has height (in inches) of density fx(x) = 1/8 e-x/8 for x > 0, and fx(x) = 0 otherwise. If she makes 20 such geysers, what is the rough...
-
What is Photosynthesis?
-
Based on the following information for the year ended December 31, 2018, what is Earnings Per Share (in dollars and cents)? Retained Earnings $ 340,000 January 1, 2018 Cost of Goods Sold $ 620,000...
-
On January 1, 2015, Y. Knott Inc. issued $100,000 in bonds payable with an interest coupon of 10%. Since the market rate of interest was close to 12%, Y. Knott had to issue the bonds at 94 (that is,...
-
Jerald earned net income of $1,200,000 in 2024. There are 950,000 shares of common stock outstanding the entire year. The preferred shareholders received a $80,000 cash dividend. Compute Jerald's...
-
On January 1, 2023, Vinson Inc. had a retained earnings balance of $60,000. The net income for the year was $110,000. he company paid dividends of $14,000 cash dividends. Prepare a December 31, 2023...
-
Find the partial fraction decomposition for each rational expression. 5 2x (x + 2)(x 1)
-
A report from the college dean indicates that for the previous semester, the grade distribution for the Department of Psychology included 135 As, 158 Bs, 140 Cs, 94 Ds, and 53 Fs. Determine what kind...
-
Provide a mechanistic explanation for each of the following reactions: (a) (b) (c) (d) acid (cat.) OH (major product) acid (cat.) (major product) AgNO3 EtOH (major product) Ph. H EtONa EtOH, ,Ph Ph...
-
Caryophyllene, a compound found in oil of cloves, has the molecular formula C15H24 and has no triple bonds. Reaction of caryophyllene with an excess of hydrogen in the presence of a platinum catalyst...
-
Squalene, an important intermediate in the biosynthesis of steroids, has the molecular formula C30H50 and has no triple bonds. (a) What is the index of hydrogen deficiency of squalene? (b) Squalene...
-
The following are extracted from the Cash Book of Jungle Trading and its Bank Statement (received from ABC Bank) for the month of April 2020: Cash Book April 2020 RM April 2020 1 Bal b/d 1,500 9...
-
1. Briefly describe FIVE (5) differences between qualitative data and quantitative data. 2. The literature review is an integral part of the research process and make a valuable contribution to...
-
Writing a detailed written assessment that challenges the traditional legal regimes governing the use of military force, addressing key jus ad bellum and jus in bello legal concerns along with their...

Study smarter with the SolutionInn App


