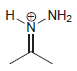
Draw all lone pairs on each of the nitrogen atoms in the compounds below. First, review in
Question:
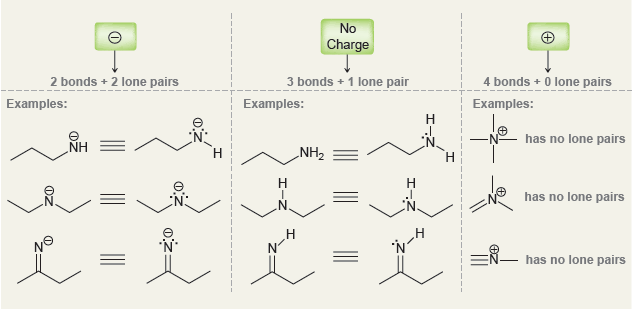
a.
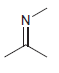
b.
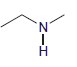
c.
d.
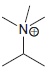
e.
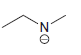
f.
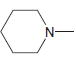
g.
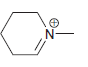
h.
Transcribed Image Text:
No Charge 3 bonds + 1 lone pair 2 bonds + 2 lone pairs 4 bonds + 0 lone pairs Examples: Examples: Examples: н has no lone pairs NH NH2 has no lone pairs .N' has no lone pairs z:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
a b c d N...View the full answer

Answered By

Muhammad Khurram
I have strong General Management skills to apply in your projects. Over last 3 years, I have acquired great knowledge of Accounting, Auditing, Microsoft Excel, Microsoft PowerPoint, Finance, Microsoft Project, Taxation, Strategic Management, Human Resource, Financial Planning, Business Planning, Microsoft Word, International Business, Entrepreneurship, General Management, Business Mathematics, Advertising, Marketing, Supply Chain, and E-commerce. I can guarantee professional services with accuracy.
4.80+
249+ Reviews
407+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw all lone pairs on each of the oxygen atoms in the compounds below. Before doing this, review in the following table, and then come back to these problems. Try to identify all lone pairs without...
-
The azide ion, N3-, is linear with two N-N bonds of equal length, 1.16 Ã. (a) Draw a Lewis structure for the azide ion. (b) With reference to Table 8.5, is the observed bond length consistent...
-
The following compound has three nitrogen atoms: Each of the nitrogen atoms exhibits a lone pair that can function as a base (to abstract a proton from an acid). Rank these three nitrogen atoms in...
-
If the owner of a company takes merchandise for personal use, what account is debited? a. Owners capital b. Owners withdrawals c. Purchases d. Cash
-
Per the Kiobel case, why might one disagree with the Court's Majority decision? Persuasively and rigorously explain.
-
Suppose a charged particle is located at the origin. What is the direction of the electric field at the point \((0.6,1.2)\) if the particle is \((a)\) positively charged and \((b)\) negatively...
-
Assuming a chain growth probability of 0.8, calculate the mole yields for hydrocarbons with 1-24 carbon atoms using the ASF distribution. How does this distribution compare to that of a 0.9 chain...
-
The income statement of Vince Gill Company is shown below. Additional information: 1. Accounts receivable decreased $360,000 during the year. 2. Prepaid expenses increased $170,000 during the year....
-
Explain the detailed relationship between money and motivation?
-
Oak Grove Inc. has a petty cash fund of $1,500. (a) Since the petty cash fund is only $1,500, should Oak Grove Inc. implement controls over petty cash? (b) What controls, if any, could be used for...
-
A carbene is a highly reactive intermediate in which a carbon atom bears a lone pair and no formal charge: How many hydrogen atoms are attached to the central carbon atom above?
-
Each of the following compounds contains both oxygen and nitrogen atoms. Identify all lone pairs in each of the following compounds: a. b. c. d. e. f. N. O=C=N
-
Suppose your company needs to raise $10 million and you want to issue 30-year bonds for this purpose. Assume the required return on your bond issue will be 9 percent, and youre evaluating two issue...
-
Calculate the missing Operating Income amount in the following multi-step income statement: Sales Revenue Cost of Goods Sold Gross Profit Operating Expenses Operating Income Interest Expense Net...
-
A firm sells $ 3 0 0 million worth of goods per year, and the average balance in accounts receivable is $ 7 5 million. ( Accounts receivable is when the firm sells an item, but hasn't collect the...
-
What are the five classification criteria that a lessee must consider to classify a non cancelable lease as a financing lease versus an operating lease. Why do you think these are important...
-
Suppose the total benefits of cement production are TB = 180q-q and the total costs of cement production are TC=q. Suppose the total external cost of cement production is TEC = 30q. 1. (2 points)...
-
A manufacturing company reports the following information. Raw materials inventory, ending Raw materials used Current Year $ 171,200 1,998,700 1 Year Ago 2 Years Ago $ 192,200 2,544,100 $ 199,200...
-
Find the partial fraction decomposition for each rational expression. 2 x2 5 + 2
-
Find the APR in each of the following cases: NUMBER OF TIMES COMPOUNDED Semiannually Monthly Weekly Infinite EAR APR 10.4% 8.9 11.6 15.4
-
Propose structures for compounds E-H. Compound E has the molecular formula C5H8 and is optically active. On catalytic hydrogenation E yields F. Compound F has the molecular formula C5H10, is...
-
Consider the interconversion of cis-2-butene and trans-2-butene. (a) What is the value of Ho for the reaction cis-2-butene : trans-2-butene? (b) Assume Ho Go. What minimum value of DG would you...
-
(a) Partial dehydrohalogenation of either (1R, 2R)-1,2-dibromo-1,2-diphenylethane or (1S, 2S)-1,2-dibromo-1,2-diphenylethane enantiomers (or a race mate of the two) produces...
-
Three people pull simultaneously on a stubborn donkey. Jack pulls directly ahead of the donkey with a force of 68.1 N, Jill pulls with 64.5 N in a direction 45 to the left, and Jane pulls in a...
-
A 1.70-kg object attached to a spring moves without friction (b = 0) and is driven by an external force given by the expression F = 4.60sin(2t), where F is in newtons and t is in seconds. The force...
-
Question 2.6 (Assessment objectives...) There are numerous support resources, services and networks available throughout Australia. They differ from state to state and support can be provided by...

Study smarter with the SolutionInn App


