Draw both conformations for each of the following compounds: (a) (b) (c) (d) (e) (f) (g) (h)
Question:
(a)
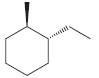
(b)
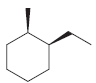
(c)
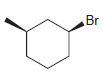
(d)
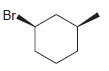
(e)
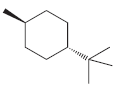
(f)
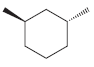
(g)
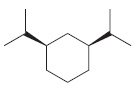
(h)
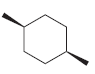
Transcribed Image Text:
Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
a b c...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw both chair conformations for each of the following compounds: (a) (b) (c) (d) (e) NH2
-
Give two names for each of the following compounds: a. b. c. d. e. f. CHCH2CHH CH CH3 CH,CH,CH CCH,CH,CH CH3CHCH2CCH2CH2CH3 CH3 CH CH2CH2CH CH3CH2CHCH2CH2CH lia CH-CHCCH-CH-CH-CH3
-
Name the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) CH,CH,C CH OH CH CH CH NO COOH Br OCH(CH)2 OH NO NO CH,OCH,CH,
-
The formula F(x) = 3x + C gives a different function for each value of C. All of these functions, however, have the same derivative with respect to x, namely F(x) = 3. Are these the only...
-
How should a corporation scan the external environment for new technological developments? Who should be responsible? What is technology research and how does it differ from market research? What is...
-
The surface tension of water in contact with air at ambient temperature is \(0.12 \mathrm{~N} / \mathrm{m}\). The pressure inside the water droplet is \(0.02 \mathrm{~N} / \mathrm{cm}^{2}\) greater...
-
A hydrogen/oxygen fuel cell operates with a voltage of \(0.7 \mathrm{~V}\) with water vapor production. Calculate the \(\mathrm{kW} / \mathrm{h}\) of work per \(\mathrm{kg}\)-mol of hydrogen and...
-
STOCK ISSUANCE (PAR, NO-PAR, AND STATED VALUE) The following stock transactions occurred during January 20-- for Bremer Corporation: (a) Issued 5,000 shares of $10 par common stock for $50,000 cash....
-
What implications would the proposal have on professionals in both public practice and industry? Do you believe an accountant in the industry should be responsible for reporting ethical violations,...
-
A balanced three phase load of 30 MW is supplied at 132 kV, 50 Hz and 0.85 lag by means of a transmission line. The series impedance of a single conductor is (20+j40) ohms and the total phase-neutral...
-
The most stable conformation of 5-hydroxy-1,3-dioxane has the OH group in an axial position, rather than an equatorial position. Provide an explanation for this observation.
-
Lindane (hexachlorocyclohexane) is an agricultural insecticide that can also be used in the treatment of head lice. Draw both chair conformations of lindane. CI CI 'CI CI I Lindane
-
Suppose that the exchange rate rose from 80 yen per U.S. dollar to 90 yen per U.S. dollar. What is the effect of this change on the quantity of U.S. dollars that people plan to sell in the foreign...
-
Why might an organization consider a remotely hosted system for HRIS?
-
Name the followings compounds and determine the empirical and molecular formulas of compounds using combustion analysis data a) Ca F b MnO4 2 FeCl3 6H0 d N2 03
-
What are the risks associated with products introductory stage?
-
Discuss the value of integrating multiple content areas to increase student engagement and achievement
-
State Poisson s and Laplace s equations and briefly describe their importance and applications
-
Find the maximum and minimum values of each objective function over the region of feasible solutions shown at the right. objective function = 3x + 5y I(1, 10) (7, 9) (7, 6) (1, 0)
-
What is the shape of the exponential distribution?
-
Given that the pKa of H2SO4 is -9 and that of HNO3 is -1.4, explain why nitration occurs more rapidly in a mixture of concentrated nitric and sulfuric acids than in concentrated nitric acid alone.
-
The following chlorides (Ph = phenyl) undergo solvolysis in ethanol at the relative rates given in parentheses. How can you explain these results? Ph Ph Ph (3 x 106) (0.08) (300)
-
Provide a detailed mechanism for each of the following reactions. Include contributing resonance structures and the resonance hybrid for the arenium ion intermediates. (a) (b) (c) HNO HSO NO2 Br Bra,...
-
Create a segmentation, selection and focus model that allows the PUMA company in Mexico to determine its customer, where to find it and how to generate value given its model. 7) Selection of the most...
-
Determine if the following values are zeroes polynomial. 4 3 f(x) = x + x-18x - 16x + 32 x+1, +2, +4 -
-
Cali Windows is a small company that installs windows. Its cost structure is as follows: (Click the icon to view the cost structure.) Calculate (a) the breakeven point in units and revenues and (b)...

Study smarter with the SolutionInn App


