The most stable conformation of 5-hydroxy-1,3-dioxane has the OH group in an axial position, rather than an
Question:
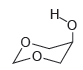
Transcribed Image Text:
Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Although the OH group is in an ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the most stable conformation of: (a) ethylcyclohexane (b) 3-isopropyl-1,1-dimethylcyclohexane (c) cis-1-tert-butyl-4-isopropylcyclohexane
-
The most stable conformation of trans-1,3-bis(1,1-dimethylethyl(cyclohexane is not a chair. What conformation would you predict for this molecule? Explain.
-
For most ketones, hydrate formation is unfavorable, because the equilibrium favors the ketone rather than the hydrate. However, the equilibrium for hydration of hexafluoroacetone favors formation of...
-
Use lHpitals rule to find the limit. 31 + 3 43 14t - t + 3 lim
-
You will work on how to receive and incorporate feedback. Specifically, you will use the feedback you received on your literature review draft
-
Go to the IRS website (www.irs.gov) and redo Problem 15, using the most recent interactive Form 4562, Depreciation and Amortization. Print out the completed Form 4562.
-
A off-axis tensile test (Figure 2.14) of a unidirectional AS/3501 carbon/epoxy specimen is conducted with \(\theta=45^{\circ}\) and the applied stress is found to be \(\sigma_{x}=15.44...
-
Overhead variances, service sector Meals on Wheels (MOW) operate a meal home-delivery service agreements with 20 restaurants to pick up and deliver meals to customers who phone or fax to MOW. MOW...
-
An ASTM A572 Grade 50 steel bar is attached to a rigid wall at one end and a spring at the other end, with the other end of the spring being attached to a rigid wall, as shown below. The bar is 20 cm...
-
During 2017, Kew Company, a service organization, had $200,000 in cash sales and $3,000,000 in credit sales. The accounts receivable balances were $400,000 and $485,000 at December 31, 2016 and 2017,...
-
Consider the following chair conformation of bromocyclohexane: (a) Identify whether the bromine atom occupies an axial position or an equatorial position in the conformation above. (b) Draw a...
-
Draw both conformations for each of the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) Br
-
In stretching a spring in an experiment, a student inadvertently stretches it past its elastic limit; the force-versus-stretch graph is shown in Fig. 5.30. Basically, after it reaches its limit, the...
-
The methodology used in your organization calls for change requests to be considered by a change control board (CCB). After some reflection and a discussion with the programmer, you have decided to...
-
Which of the following describes the purpose of a strategy map? a. A graphical description of expected cause-and-effect linkages among Balanced Scorecard perspectives b. A list of the companys...
-
The Swatch Group (swatchgroup.com) with its headquarters in Biel, Switzerland, is a world-renowned manufacturer of watches and jewelry. The company was founded in 1983 by Lebanese-born Nicolas Hayek...
-
The CEO of your organization was so impressed with your last project that you have been given responsibility with a larger, even more important project. The CEO calls you in for a discussion...
-
You are the project manager for a company that is building a behavioral health system for some of the counties in your state. The project is slightly ahead of schedule and there havent been any...
-
Draw a utility function that exhibits risk-loving behavior for small gambles and risk-averse behavior for larger gambles.
-
Why did management adopt the new plan even though it provides a smaller expected number of exposures than the original plan recommended by the original linear programming model?
-
Account for the following observations with mechanistic explanations. (a) At high concentration of ethoxide, the rate depends on both the allylic halide and ethoxide concentrations. (b) At low...
-
1-Chloro-3-methyl-2-butene undergoes hydrolysis in a mixture of water and dioxane at a rate that is more than a thousand times that of 1-chloro-2-butene. (a) What factor accounts for the difference...
-
Primary halides of the type ROCH2X apparently undergo SN1-type reactions, whereas most primary halides do not. Can you propose a resonance explanation for the ability of halides of the type ROCH2X to...
-
How is decision-making in organizations best supported by Management Accounting (a descriptive nature) and how should it be supported by Management Accounting (a prescriptive nature)? Is there a gap...
-
Discuss the thermodynamic principles governing the separation of azeotropic mixtures using advanced distillation techniques such as pressure swing distillation, extractive distillation, and hybrid...
-
respond to the discussion In the IMA article, Activity-Based Costing (ABC) is explained as a cost allocation technique that traces indirect costs (often referred to as "overhead") to specific cost...

Study smarter with the SolutionInn App


