Draw the major product(s) that are expected when each of the following amines is treated with excess
Question:
(a)
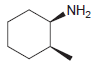
(b)
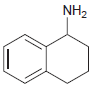
Transcribed Image Text:
NH2 NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)

Answered By

Vincent Omondi
I am an extremely self-motivated person who firmly believes in his abilities. With high sensitivity to task and operating parameters, deadlines and keen on instructions, I deliver the best quality work for my clients. I handle tasks ranging from assignments to projects.
4.90+
109+ Reviews
314+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products that are expected when each of the following alkenes is treated with a peroxy acid (such as MCPBA) followed by aqueous acid: a. b. c. d. e. f.
-
Predict the products that are expected when each of the following alkenes is treated with ozone followed by DMS: a. b. c. d. e. f.
-
What product(s) are expected when each of the following compounds reacts with one equivalent of NBS in CC14 in the presence of light and peroxides? Explain your answers. (a) cyclohexene (b)...
-
In Fig. P2.72 gate AB is circular. Find the moment of the hydrostatic force on this gate about axis A. Neglect atmospheric pressure.
-
Find the sum of the finite geometric sequence. 1. 2. 3. 4. -1 2i i=1 31 i=1
-
Consider a typical diverging passenger-side mirror with a focal length of -80 cm. A 1.5-m-tall cyclist on a bicycle is 25 m from the mirror. You are 1.0 m from the mirror, and suppose, for...
-
Find the binary pair diffusivity for the system methane (A)-ethane (B) at \(293 \mathrm{~K}\) and \(1 \mathrm{~atm}\) by using the Lennard-Jones method given by Eq. (1.53) by the following methods....
-
Outdoor Living has the following accounts receivable at year end, broken down by age: Prior experience has shown that the company will probably collect 95% of its current receivables. Furthermore,...
-
Suppose you have $1,000.00 and face two alternatives: 1. Put the money in an interest-bearing account that pays 0.5% monthly. This account is secured against bank failure. 2. Hold the money in cash....
-
The frame supports a centrally applied distributed load of 1.8 kip>ft. Determine the state of stress at points A and B on member CD and indicate the results on a volume element located at each of...
-
Consider the structure of the azo dye called alizarine yellow R (below). Show the reagents you would use to prepare this compound via an azo coupling process. .N. N' O2N
-
If Davis adds a line of apparel or shoes, should he try to sell his line to other retail clothing shops in Calgary or elsewhere in Canada?
-
Balance the following equations: (a) Al 4 C 3 (s) + H 2 O(l)Al(OH) 3 (s) + CH 4 (g) (b) C 5 H 10 O 2 (l) + O 2 (g)CO 2 (g) + H 2 O(g) (c) Fe(OH) 3 (s) + H 2 SO 4 (aq)Fe 2 (SO 4 ) 3 (aq) + H 2 O(l)...
-
Which type of organization design best fits an uncertain and changing environment? (a) Mechanistic (b) Bureaucratic (c) Organic (d) Traditional
-
Faisal Sham supervises a group of seven project engineers. His unit is experiencing a heavy workload, as the demand for different versions of one of his firms computer components is growing. Faisal...
-
For the current month, Jackson Cement Co. incurred payroll expenses as follows: a. What amount can Jackson claim as a tax deduction for salary and wage expense? b. How much can Jackson deduct as tax...
-
When a new business is quick to act and captures a market niche before competitors, this is called____________. (a) intrapreneurship (b) an initial public offering (c) succession planning (d)...
-
Planning is the process of __________ and __________. (a) developing premises about the future, evaluating them (b) measuring results, taking corrective action (c) measuring past performance,...
-
What is vector addition and how is it done?
-
QUESTION 2 The CEO of Farisha Hijab Sdn Bhd insisted on further investigation to be carried out that he also required Mr Muaz to conduct the analysis of variance for the material and labour of the...
-
Predict which compound in each pair has the higher boiling point. Explain your prediction. (a) CH3CH2OCH3 or CH3CH1OH2CH3 (b) CH3CH2CH2CH3 or CH3CH2CH2CH2CH3 (c) CH3CH2CH2CH2CH3 or (CH3)2CHCH2CH3 (d)...
-
Circle the functional groups in the following structures. State to which class (or classes) of compounds the structure belongs. (a) (b) (c) (d) (e) (f) (g) (h) (i) COOH N--H CH2OCH3 CN NH CH3 CHCOOCH...
-
Dimethyl sulfoxide (DMSO) has been used as an anti-inflammatory rub for race horses. DMSO and acetone appear to have similar structures, but the C=O carbon atom in acetone is planar, while the S=O...
-
Explain how the OS and Utility programs work with application software. Summarize the features of several embedded operating systems course: introduction to information technology code: EBI...
-
Please explain and describe what are input and output devices. Explain what the differences are between an operating system, an embedded operating system, and a network operating system. Explain and...
-
Determine the complexity of the following pseudocode snippets in Big-O and Big-Q2. Do these code snippets have a Big-e? What are the functions doing? 1) my_func(some_nums) result = 0 for (num in...

Study smarter with the SolutionInn App


