Each compound below exhibits one lone pair. In each case, identify the type of atomic orbital in
Question:
a.
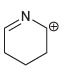
b.
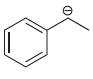
c.
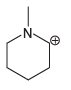
Transcribed Image Text:
.N.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
a An sp 2 hy...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine whether each compound below exhibits a molecular dipole moment: a) CH 4 b) NH 3 c) H 2 O d) CO 2 e) CCl 4 f) CH 2 Br 2
-
For each compound below, identify all lone pairs and indicate whether each lone pair is localized or delocalized. Then, use that information to determine the hybridization state and geometry for each...
-
The compound 1,1,1-trifl uoroacetylacetone (tfa) is a bidentate ligand: It forms a tetrahedral complex with Be2+ and a square planar complex with Cu2+. Draw structures of these complex ions and...
-
In Exercises find the second derivative of the function. f(x) = x + 3x-3
-
On August 1, Clayton Co. issued 1,300,000 of 20 year, 9% bonds, dated August 1, for $1,225,000. Interest is payable semiannually on February 1 and August 1. Present the entries to record the...
-
A real estate company operates 25 offices scattered around the southeastern United States. Each office employs six agents. Each month, the CEO receives a summary report of the average value of home...
-
The table below reports the unemployment rate, labor force participation rate, and (working-age) population for the United States in January 2008, 2011, and 2016. Using the data, answer the following...
-
Cosi Company reported net income of $190,000 for 2014. Cosi also reported depreciation expense of $35,000 and a loss of $5,000 on the disposal of plant assets. The comparative balance sheet shows an...
-
A steam turbine having an isentropic turbine efficiency of 90% drives an air compressor having an isentropic compressor efficiency of 85%. Steady-state operating data are provided on the figure....
-
The information below relates to questions 1- 11. Emily and Sibusisiwe are two sisters of the Nkoane family who have recently qualified as chartered accountants. The two sisters are in a partnership,...
-
Learning to extract structural information from molecular formulas: a) Write out the molecular formula for each of the following compounds: Compare the molecular formulas for the above compounds and...
-
Draw all significant resonance structures for each of the following compounds: a. b. c. N' z-
-
Given the linked representation of a pure list such as write an in-place reversal algorithm to reverse the sublists at all levels including the topmost level. For this example, the result would be a...
-
What is transfer prices? What are the different types of transfer prices? Under what circumstances is each type of transfer price appropriate and useful?
-
What kind of performance is measured in profit centres? What are the criteria for evaluating that performance?
-
Explain why a decision centre should be treated as a profit centre rather than as a cost centre.
-
Define an expense centre. What is the suitability of the measure of performance in an expense centre?
-
The management control process involves human beings, from those in the lowest responsibility centres of the organisational hierarchy up to and including each member of top management. Discuss.
-
How does XML differ from HTML?
-
Find a polar equation for the curve represented by the given Cartesian equation. 4y 2 = x
-
Using the method of Section 9.8A, determine the number of expected signals for the following compounds. (a) (b) (c) CH3
-
Compound S (C8H16) reacts with one mole of bromine to form a compound with molecular formula C8H16Br2. The broadband proton-decoupled 13C spectrum of S is given in Fig. 9.49. Propose a structure for...
-
A compound with molecular formula C4H8O has a strong IR absorption at 1730 cm-1. Its mass spectrum includes key peaks at m/z 44 (the base peak) and m/z 29. Propose a structure for the compound and...
-
What are the vulnerabilities of embedded operating systems, such as, but not limited to, IoT devices, programmable logic devices, and vehicle control systems.
-
Factor the polynomial completely. 7x-17x-12
-
What are some relevant recommendations about Coca-Cola's Planning, Design, and Implementation of an Enterprise Resource Planning System that other companies can apply to their own initiatives?

Study smarter with the SolutionInn App


