For each compound below, identify all lone pairs and indicate whether each lone pair is localized or
Question:
a.
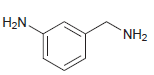
b.
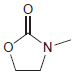
c.
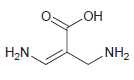
d.
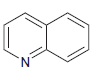
e.
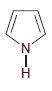
f.
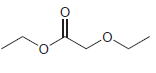
Transcribed Image Text:
H2N `NH2 z.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
a b c d e f H IZ delo...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw all lone pairs on each of the oxygen atoms in the compounds below. Before doing this, review in the following table, and then come back to these problems. Try to identify all lone pairs without...
-
Draw all lone pairs on each of the nitrogen atoms in the compounds below. First, review in the following table, and then come back to these problems. Try to identify all lone pairs without having to...
-
Each of the following compounds contains both oxygen and nitrogen atoms. Identify all lone pairs in each of the following compounds: a. b. c. d. e. f. N. O=C=N
-
Compute the indicated quantities for the given homomorphism. Ker () for : S 3 Z 2 in Example 13.3 Data from Example 13.3 Let S n be the symmetric group on n letters, and let : S n Z 2 be defined by...
-
You have been hired by First Citizens Bank as a financial analyst. One of your first job assignments is to analyze the present financial condition of Bradley Stores, Incorporated. You are provided...
-
Some equipotential lines surrounding a negatively charged object are shown in Figure P25.26, where the potential difference between any two adjacent lines is the same. (a) In which region is the...
-
What are the advantages and limitations of performance indicators to be used in a National Health Service hospital trust? Provide examples of areas where they can be used.
-
During 2014, Fargo Company purchased land for $375,000. It paid $125,000 in cash and signed a $250,000 mortgage for the rest. The company also sold for $95,000 cash a building that originally cost...
-
Mauve Company uses a standard cost system that applies manufacturing overhead to units of product on the basis of direct labour hours (DLHS). The following data pertain to last month: Actual Hours...
-
The Off-Campus Playhouse adjusts its accounts every month. The following is the company's unadjusted trial balance dated September 30, current year. Additional information is provided for use in...
-
Use LHpitals rule lim [f(x)/g(x)] x 0 = lim [df(x)dx/dg(x)/dx] x 0 to show that the expression derived for Pf in part b of Example problem 1.1 hane the correct limit as y 0.
-
Nicotine is a toxic substance present in tobacco leaves. There are two lone pairs in the structure of nicotine. In general, localized lone pairs are much more reactive than delocalized lone pairs....
-
Determine the appropriate functions. Express the cost C of insulating a cylindrical water tank of height 2 m as a function of its radius r, if the cost of insulation is $3 per square meter.
-
Q1. Explain How you would identify an organization's standards and values when they are: a.Explicitly stated? b. Implied in the way business is conducted?
-
Peifan is taking an Interest Only loan to purchase a $669,425 property with 0.9 LTV. The loan is issued for 15 years with monthly payments and has a fixed interest rate at 5%. Under this mortgage...
-
Rainy State Government had good revenues and generated enough surplus to invest in $180,000 shares issued by North Bank, an Australian commercial bank (transaction 1). Hobby Club, which had...
-
The S&P 500 Index closed at 2863.39 on April 28 and closed at 2939.51 on April 29. Calculate the continuously compounded return on the index from April 28 to April 29.Answer to the nearest hundredth...
-
If the risk rate of the return is 4%, the return on the market portfolio is 12%, the beta of the hotel norwood is 1.5, what is the expected rate of the return for hotel norwood?
-
Work each problem. Prove that any square matrix has no more than one inverse.
-
The maximum pressure that can be developed for a certain fluid power cylinder is 15.0 MPa. Compute the required diameter for the piston if the cylinder must exert a force of 30 kN.
-
Outline all steps in a mechanism showing how 2, 3-dimethyl-2-butanol is formed in the acid-catalyzed hydration of 3, 3-dimethyl-1-butene.
-
Alkyl halides add to alkenes in the presence of AlCl3; yields are the highest when tertiary halides are used. Predict the outcome of the reaction of tert-pentyl chloride (1-chloro-2,...
-
Explain the stereochemical results observed in this catalytic hydrogenation. (You may find it helpful to build hand-held molecular models.) CH2 CH CH3 CH Pt catalyst CH3 HOAc 170%) 11 (30%)
-
When owning and operating any business, whether it's a caf or a pool cleaning business, it's very important that you know exactly what your income and expenditure is. Is your business making money or...
-
Resources Unlimited Corporation was formed in 1985 through the merger of two natural pipeline companies. The result was the largest gas distribution network in the United States, with 38,000 miles of...
-
Toyota was the world's first automobile manufacturer to produce more than 10 million vehicles per year, which it has done since 2012, when it also reported the production of its 200-millionth...

Study smarter with the SolutionInn App


