For each of the following compounds, identify the expected chemical shift for each type of proton: (a)
Question:
(a)
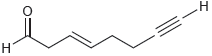
(b)
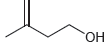
(c)
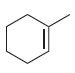
(d)
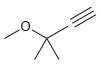
Transcribed Image Text:
н н ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)
a b c d 10 pp...View the full answer

Answered By

Mario Alvarez
I teach Statistics and Probability for students of my university ( Univerisity Centroamerican Jose Simeon Canas) in my free time and when students ask for me, I prepare and teach students that are in courses of Statistics and Probability. Also I teach students of the University Francisco Gavidia and Universidad of El Salvador that need help in some topics about Statistics, Probability, Math, Calculus. I love teaching Statistics and Probability! Why me?
** I have experience in Statistics and Probability topics for middle school, high school and university.
** I always want to share my knowledge with my students and have a great relationship with them.
** I have experience working with students online.
** I am very patient with my students and highly committed with them
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw both chair conformations for each of the following compounds. In each case, identify the more stable chair conformation: (a) Methylcyclohexane (b) Trans-1,2-Diisopropylcyclohexane (c)...
-
For each of the following compounds, identify any polar covalent bonds by drawing δ+ and δ- symbols in the appropriate locations. a. b. c. d. e. f. -- :F: - : CI
-
For each of the following compounds, identify whether each C R C system is cumulated, conjugated, or isolated: (a) (b) (c) (d) HO HO- cis-Aconitic acid Plays a role in the citric acid cycle
-
Swish Designs specialises in designing commercial office space in Auckland. The CEO, Ralph Polo has reviewed the financial results and has noticed that operating profits were below budget. He also...
-
Explain the role of inflationary expectations, economic contraction, and Federal Reserve Policy in producing the record low interest rates observed at the end of 2007 and the beginning of 2008.
-
Mechanistic hypothesis and efficient market hypothesis Hudson Ltd had always classified interest paid as an operating cash flow. In 2024 Hudson Ltd changed its accounting policy and classified...
-
Which of the following is an extensive property? (a) Free energy (c) Specific heat (b) Refractive index (d) Surface tension.
-
Emil Skoda Company had the following adjusted trial balance. Instructions(a) Prepare closing entries at June 30, 2010.(b) Prepare a post-closing trialbalance. EMIL SKODA COMPANY Adjusted Trial...
-
Davis Depot invested $12,600 in a savings account paying 4% interest on January 1, 2025, to accumulate funds to buy a new forklift on January 1, 2030. What amount will be in the savings account as of...
-
Richard chooses technique 0 and 2 requiring 10+10-20 efforts and provising 10+11=21 benefits. Hence, 21 is returned as the output Example 2: input1: 3 input2: (10,10,10,10) input3: (10,11,12,15)...
-
What data are necessary to construct a financial break-even chart?
-
A compound with molecular formula C 5 H 10 O 2 has the following NMR spectrum. Determine the number of protons giving rise to each signal. Proton NMR lle 2.0 1.5 0.5 ppm 4.5 4.0 33.2 3.5 3.0 2.5 1.0...
-
In Exercises find the center of mass of the point masses lying on the x-axis. m = 8, m = 5, m3 = 5, m = 12, m5 = 2 ms 0, x X3 x4 X5 = 3, x X = -2, x = 6, x3 = = -5
-
Name three ( 3 ) efficient methods that can be used for the verification of accuracy and reliability of property data.
-
List two reasons why it s important to keep records of risk assessments.
-
Analyze the Trivago s search engine marketing strategy ( search engine optimization and paid search marketing ).
-
If a time-series is not stationary, how can stationarity of that series be obtained? (1 mark) Explain the relationship between order of integration and non-stationary. (3 marks) Explain the...
-
Knowing last weeks weather conditions will not help when trying to decide what coat to wear today" - Is an example of which characteristic of high - quality Data / information ?
-
Perform the operation mentally. (0.25 3 )(400 3 )
-
Which of the companies has the lowest accounts receivable turnover in the year 20X2? a. Company A. b. Company B. c. Company C. d. CompanyD. 20X1 20X2 Credit Sales Average Receivables Balance $1.0...
-
Draw the most stable conformation of 1, 4-dichiorobutane, using wedges and dashes to represent bonds coming out of the paper and going behind the paper, respectively.
-
Malic acid, C4H6O5, has been isolated from apples. Because this compound reacts with 2 molar equivalents of base, it is a dicarboxylic acid. (a) Draw at least five possible structures. (b) If malic...
-
Formaldehyde, H2C = O, is known to all biologists because of its usefulness as a tissue preservative. When pure, formaldehyde trimerizes to give trioxane, C3H6O3, which, surprisingly enough, has no...
-
An object is moving with an initial velocity of 4 0 m / s west. If it has a constant acceleration of 6 m / s ^ 2 due east, what is its velocity after 5 seconds? How long did it move at that time?
-
Haliburton Mills Inc. is a large producer of men's and women's clothing. The company uses standard costs for all of its products. The standard costs and actual costs for a recent period are given...
-
A block with a mass of 11 kg is placed on a inclined plane with an angle of inclination of 45 degrees. The coefficient of kinetic friction between the block and the surface is 0 . 2 . a . Calculate...

Study smarter with the SolutionInn App


