For each of the following compounds, predict the number of signals and location of each signal in
Question:
(a)
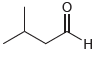
(b)
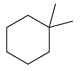
(c)
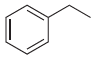
(d)
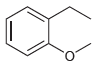
(e)
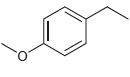
(f)
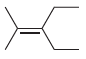
(g)
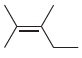
(h)
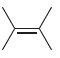
(i)
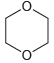
(j)
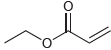
Transcribed Image Text:
H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (18 reviews)
a Four signals Three appear in the region 0 50 ppm and the fourth signal the CO appears in the regio...View the full answer

Answered By

Kainat Shabbir
i am an experienced qualified expert with a long record of success helping clients overcome specific difficulties in information technology, business and arts greatly increasing their confidence in these topics. i am providing professional services in following concerns research papers, term papers, dissertation writing, book reports, biography writing, proofreading, editing, article critique, book review, coursework, c++, java, bootstarp, database.
5.00+
184+ Reviews
255+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many 1H NMR signals (not peaks) would you predict for each of the following compounds? (Consider all protons that would be chemical shift nonequivalent.) (a) (b) (c) (d) (e) (f) (g) (h) (i)
-
Identify the molecular formula for each of the following compounds, and then predict the mass of the expected molecular ion in the mass spectrum of each compound. a. b. c. d. e.
-
For each of the following compounds, 1. Draw the Lewis structure. 2. Show how the bond dipole moments (and those of any nonbonding pairs of electrons) contribute to the molecular dipole moment. 3....
-
An important U.S. government organization charged with setting human resource management guidelines is O the EEOC (Equal Employment Opportunity Commission). the OSHA (Occupational Safety and Health...
-
What are two reasons for the failure of an EIS?
-
Here are the residuals for a regression of Sales on Number of Sales People Working for the bookstore of Exercise 1: a) What are the units of the residuals? b) Which residual contributes the most to...
-
What are the various types of refrigerant?
-
Horse Country Living publishes a monthly magazine for which a 12-month subscription costs $30. All subscriptions require payment of the full $30 in advance. On August 1, 2010, the balance in the...
-
On 8 February 19X8 PJ, a London trader, consigned 120 cases of goods to MB, an agent in New Zealand. The cost of the goods was 25 a case. PJ paid carriage to the port 147 and insurance 93. On 31...
-
Flynn Fireballer has been playing baseball since he was five years old and has always dreamed of playing in the big leagues. Last season, he was a starting pitcher for a double-A (AA)-level baseball...
-
Workers are compensated by firms with benefits in addition to wages and salaries. The most prominent benefit offered by many firms is health insurance. Suppose that in 2000 workers at one steel plant...
-
Explain why the effective interest rate (r EAR ) on a loan often is much higher than the simple, or stated, interest rate (APR).
-
A machine in the student lounge dispenses coffee. The average cup of coffee is supposed to contain 7.0 ounces. Eight cups of coffee from this machine show the average content to be 7.3 ounces with a...
-
Explain the factors in the external environment that have led to the increased interest in strategic reward management;
-
Outline the strengths and limitations for diversity management of the equal opportunities approach.
-
What do you consider to be the strengths and limitations of the equal opportunities and the managing diversity approaches to diversity management?
-
The Tibbett and Britten Group (TBG) is a UK-based international logistics service provider. Its main customers are retailers and manufacturers, to whom it provides warehouse, distribution and...
-
Outline how perceptions of distributive, procedural, informational and interpersonal treatment may impact positively on downsizing survivors reactions.
-
In 2018, Gale and Cathy Alexander hosted an exchange student, Axel Muller, for 9 months. Axel was part of International Student Exchange Programs (a qualified organization). Axel attended tenth grade...
-
Prove the following D,(cos x) = - sin x (Hint: Apply the identity cos(A + B) = cos A cos B sin A sin B)
-
Reaction of 2-butanone with HCN yields a chiral product. What stereochemistry does the product have? Is it optically active?
-
How would you synthesize the following compounds from Cyclohexanone? (a) 1-Methylcyclohexane (b) 2-Phenylcyclohexanone (c) cis-1, 2-Cyclohcxanediol (d) 1-Cyclohexylcyclohcxanol
-
One of the steps in the metabolism of fats is the reaction of an unsaturated acyl CoA with water to give a -hydroxy-acyl CoA. Propose a mechanism.
-
A one cubic foot sample of a borrow-source clay weighed 88 lbs. If the specific gravity of solids was measured as 2.70, and the clay was found to be 10 percent saturated, determine the water content...
-
Evaluate the use of novel catalytic systems, such as structured catalysts, catalytic distillation, and multifunctional catalysts, in achieving process intensification, discussing the effects on...
-
Find the minimum tractive effort required for vehicle to maintain 70mph speed at 5%upgrade through an air density of 0.002045 slug/ft^3. Show all steps and unit conversion please Problem 2:...

Study smarter with the SolutionInn App


