For each of the following pairs of compounds determine whether the two compounds would be at equilibrium
Question:
For each of the following pairs of compounds determine whether the two compounds would be at equilibrium at room temperature or whether they could be isolated from one another:
(a)
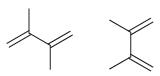
(b)
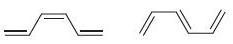
(c)
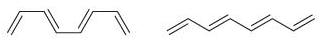
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a These drawings represent two different conformations of the same compound the scis conformation ...View the full answer

Answered By

Raunak Agarwal
Teaching is my hobby and now my profession. I teach students of CA and CFA(USA) in batches of 100 students and have a 5 year experience.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following pairs of compounds, determine whether they are enantiomers, epimers, diastereomers that are not epimers, or identical compounds: (a) (b) (c) (d) . O: - - - - - - - -...
-
For each of the following pairs of compounds, determine whether they are enantiomers, epimers, diastereomers that are not epimers, or identical compounds: (a) d-Glucose and d-gulose (b)...
-
For each of the following pairs of compounds, determine which compound is more stable (you may find it helpful to draw out the chair conformations): (a) (b) (c) (d) II
-
Common structural steel can be represented on drawings using a letter, immediately followed by a number, then another number (for example, S14x30). The two numbers for a steel member W16x60 designate...
-
A sub-rounded to sub-angular sand has a D10 of about 0.1 mm and a uniformity coefficient of 3. The angle of shearing resistance measured in the direct shear test was 47. Is this reasonable? Why or...
-
How do social identities, power differentials, and poor structural integration affect the successful management of diversity?
-
A safety PLC has an Ethernet connection port used to send information to a data server in the system. Can this port be used without a cybersecurity risk?
-
Webber fabricating estimated the following annual costs. Expected annual direct labor hours................ 40,000 Expected annual direct labor cost.............. $ 625,000 Expected machine...
-
Disney needs to close Magic Kingdom to remodel and update the castle. Disney's advisors presented two renovation alternatives: (1) a quick facelift or (2) a complete rebuild. The quick facelift would...
-
Patrons at a local restaurant were asked to rate their recent experience at the restaurant with respect to its advertised atmosphere of upbeat, comfortable, and clean. Possible responses included...
-
Diamond Hill Jewelers is considering the following independent projects: Which project(s) should be accepted if the required rate of return for the projects is 10 percent? Compute the NPV and the IRR...
-
What is the principle of competitive exclusion? Of resource partitioning?
-
The US is the only Western first-world nation that still uses the death penalty. This has caused some friction between the US and EU nations; for example, a recent ban on lethal injection drugs from...
-
Jeff Jones is a new pharmacy student at the state college located in your town. You (as pharmacist in charge) hire Jeff to be a pharmacy intern. While filling out his intern papers, Jeff notices that...
-
Mrs. Smith, a long time patient at your pharmacy, went on vacation to another country. While on her vacation, she realized she forgot her overthe-counter pain medication and visited a pharmacy to...
-
Jim is a pharmacist who works for a pharmacy chain in a large city. Jim developed a business that illegally solicited bets on sporting events. He promoted this to patients, and several patients would...
-
Lacey has been a pharmacist at a local chain pharmacy, Uptown Pharmacy, for 2 months. Lacey received a few days of training once she started; however, she has made a few dispensing errors. One error...
-
The board of pharmacy can take action against a licensee only when the licensee has been given notice of the action and an opportunity to explain the licensees side of the story at some sort of...
-
Maggie is single and supports her 85-year-old parents who have no income and live in a home rented for them by Maggie. What is Maggies filing status and why?
-
Currently, there are five concepts of food stands, including: hot dogs, soft pretzels, turkey legs, sandwich wraps, and funnel cakes. This approach will double the existing number of food stands...
-
Outline methods for the preparation of acetophenone (phenyl methyl ketone) starting from the following: (a) Benzene (b) Bromobenzene (c) Methyl benzoate (d) Benzonitrile (e) Styrene
-
The following reactivity order has been found for the basic hydrolysis of p-substituted methyl benzoates: Y = NO 2 > Br > H > CH 3 > OCH 3 . How can you explain this reactivity order? Where would you...
-
The following reactivity order has been found for the saponification of alkyl acetates by aqueous NaOH. Explain. CH3CO2CH3 > CH3CO2CH2CH3 > CH3CO2CH (CH3)2 > CH3CO2C (CH3)3
-
There are 42 locations throughout the northeast region with deposits totaling $3.2bil. If you were Kiamarie's vice president, how would you expect her to choose between Gagne's Nine Events of...
-
Why is succession planning essential in an organization? 2. What is the need for succession planning? 3. What do you think are the challenges to succession planning? 4. How can employees contribute...
-
Two activities/obligations/responsibilities that might be added to an updated version of the job description for an HR professional: How would you go about monitoring changes in laws and regulatory...

Study smarter with the SolutionInn App


