For each of the following processes predict the sign of ÎS for the reaction. In other words,
Question:
a.
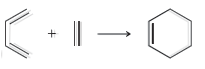
b.
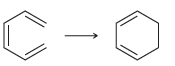
c.
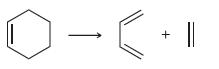
d.
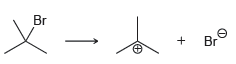
e.
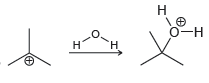
f.
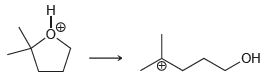
Transcribed Image Text:
||
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a S sys is expected to be negative a decrease in entropy because tw...View the full answer

Answered By

ADHITHYA NARAYANAN
I have cleared competitive exams like GATE and JEST. I also have online problem solving experiences which would come good here as well. I have experience in tutoring in Chegg and BNED platforms.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the sign of ÎS accompanying this reaction. Explain your choice.
-
The process descriptions, process flow diagrams, and technical data for processes used commercially in 38 chemical industries. For each of the following processes, draw a block-flow diagram of just...
-
For each of the following processes, indicate whether the signs of S and H are expected to be positive, negative, or about zero. (a) A solid sublimes. (b) The temperature of a sample of Co(s) is...
-
Diagonalize the matrices, if possible. The eigenvalues are as follows: (11) = 1, 2, 3; (12) = 1, 4; (13) = 5, 1; (14) = 3, 4; (15) = 3, 1; (16) = 2, 1. 3 4 6 1
-
Q1) what is the role of the project manager in the selection of the project? What criteria does the project manager use to select the project, and how are these criteria derived? Q2) List and briefly...
-
Assume the following information for Kingbird Corp. Accounts receivable (beginning balance) $139,000 Allowance for doubtful accounts (beginning balance) 11,370 Net credit sales 944,000 Collections...
-
An orthotropic lamina has the following properties: \[E_{1}=160 \mathrm{GPa} \quad s_{\mathrm{L}}^{(+)}=1800 \mathrm{MPa}\] \[\begin{aligned} & E_{2}=10 \mathrm{GPa} \quad s_{\mathrm{L}}^{(-)}=1400...
-
At the beginning of 2011, the Healthy Life Food Company purchased equipment for $42 million to be used in the manufacture of a new line of gourmet frozen foods. The equipment was estimated to have a...
-
tes Outback Outfitters sells recreational equipment. One of the company's products, a small camp stove, sells for $110 per unit. Variable expenses are $77 per stove, and fixed expenses associated...
-
Dixon Robotics manufactures three robot models: the A3B4, the BC11, and the C3PO. Dixon allocates manufacturing overhead to each model based on machine hours. A large portion of the companys...
-
Using the data in the following table, predict the sign and magnitude of ÎH° for each of the following reactions. In each case, identify whether the reaction is expected to be endothermic...
-
At room temperature, molecules spend most of their time in lower energy conformations. In fact, there is a general tendency for any system to move toward lower energy. As another example, electrons...
-
Let have degree n. Suppose that for some k (0 n ; p a k ; p| a i for all O i k - 1; and p 2 a 0 Show that has a factor g of degree at least k that is irreducible in Z[x]. [= ax Z[x] i-0
-
The discouraged worker category is defined to include people who: A. are overqualified for their jobs. B. could look for a job but choose not to. C. currently look for work without finding it.
-
Why are the notes and supplementary information sections important parts of the financial statements?
-
Monetarists favor a limited role for the government because they argue that: A. government policies operate with a lag. B. firms take time to adjust to systemic shocks to the economy. C. resource use...
-
The inventory-to-sales ratio is most likely to be rising: A. as a contraction unfolds. B. partially into a recovery. C. near the top of an economic cycle.
-
What types of requirements have been placed on not-for-profit hospitals to ensure that they meet their charitable mission?
-
True or false? If the law of diminishing marginal product did not hold, the worlds food supply could be grown in a flowerpot.
-
Classify each of the following as direct costs or indirect costs of operating the Pediatrics ward for children at the Cleveland Clinic: a. Wi-Fi covering the entire hospital campus b. Net cost of...
-
Outline all steps in a malonic ester synthesis of each of the following: (a) pentanoic acid, (b) 2-methylpentanoic acid, (c) 4-methylpentanoic acid.
-
The antiepileptic drug valproic acid is 2-propylpentanoic acid (administered as the sodium salt). One commercial synthesis of valproic acid begins with ethyl cyanoacetate. The penultimate step of...
-
Show how you could employ enamines in syntheses of the following compounds: (a) (b) (c) (d) O C OEt
-
7. An electric charge Q is distributed uniformly throughout a nonconducting sphere of radius Ro with a uniform density, p = Q/{(4/3)R}. Use Gauss's Law to determine (a) (5pts) the electric field,...
-
A child is swinging a 330-g ball at the end of a 66.0-cm-long string in a vertical circle. The string can withstand a tension of 11.0 N before breaking. (a) What is the tension in the string when the...
-
A steel cable that weighs 8 lb/ft is used to pull a 500 lb block of concrete from the ground to the top of a 120 ft tall building. Let x be the distance, in feet, from the block to the TOP of the...

Study smarter with the SolutionInn App


