For each of the following reactions assume a stepwise process is taking place and draw the mechanism:
Question:
(a)
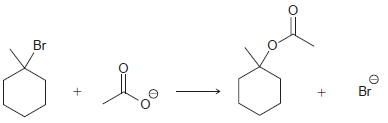
(b)

Transcribed Image Text:
Br Br NaCl CI Nal
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
a b...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following reactions, assume a concerted process is taking place and draw the mechanism: (a) (b) SH Br NaSH NaBr Nal NAOME NaOMe
-
Draw a mechanism for each of the following reactions: (a) (b) 1) EtMgBr 2) - I + HCI
-
Draw a mechanism for each of the following reactions: (a) (b) HO 1) EtMgBr 2) H20
-
A driver of a car is initially 455 miles from home, traveling toward home on a straight freeway at 70 miles per hour. (a) Write a formula for a linear function f that models the distance between the...
-
Describe the basic factors that influence the selection of a project organizational form, and their influences. Compare and contrast the role of the functional manager and the project manager in...
-
A flat belt is required to transmit \(35 \mathrm{~kW}\) from a pulley of \(1.5 \mathrm{~m}\) effective diameter running at 300 \(\mathrm{rpm}\). The angle of contact is spread over 11/24 of the...
-
In auditing the financial statements of a manufacturing company that were prepared from data processed by electronic data processing equipment, the CPA has found that his traditional "audit trail"...
-
Lynch Company manufactures and sells a single product. The following costs were incurred during the company's first year of operations: During the year, the company produced 25,000 units and 20,000...
-
On January 1, 2020, Hull Corp. paid $ 516,000 for 24% (56,000 shares) of Oliver Co outstanding common stock. Hull uses the equity method. The investment balance at the end of 2020 was $672,000. On...
-
The following classification scheme typically is used in the preparation of a balance sheet: a. Current assets b. Investments and funds g. c. Property, plant, and equipment d. Intangible assets i....
-
Assign a systematic name for each of the following compounds: (a) (b) (c) (d) CI Br
-
When a nucleophile and electrophile are tethered to each other (that is, both present in the same compound), an intramolecular substitution reaction can occur, as shown. Assume that this reaction...
-
Using the property data in Appendix D, reproduce Fig. 13.4. Note that the initial temperature and pressure are 298 K and 1 atm, respectively. Spreadsheet software is recommended to facilitate your...
-
MCO Leather manufactures leather purses. Each purse requires 2 pounds of direct materials at a cost of $5 per pound and 0.7 direct labor hour at a rate of $13 per hour. Variable overhead is budgeted...
-
how do you calcuate Unit Profit, Sales, and Profit in excel?
-
Explain how tone, structure and style of your communication may impact on others. In your answer: define tone, style and structure as they relate to communication. explain how the tone, style and...
-
Imperial Jewelers manufactures and sells a gold bracelet for $404.00. The company's accounting system says that the unit product cost for this bracelet is $267.00 as shown below: Direct materials...
-
Westex Products is a wholesale distributor of industrial cleaning products. When the treasurer of Westex Products approached the company's bank late in the current year seeking short-term financing,...
-
Use Exercise 25 to find the moment of inertia of a circular disk of radius a with constant density about a diameter. (Compare with Example 15.4.4.)
-
Refrigerant-134a enters an adiabatic compressor as saturated vapor at 120 kPa at a rate of 0.3 m3/min and exits at 1-MPa pressure. If the isentropic efficiency of the compressor is 80 percent,...
-
The first synthesis of a crown ether (Section 11.16) by C. J. Pedersen (of the DuPont Company) involved treating 1, 2-benzenediol with di(2-chloroethyl) ether, (ClCH2CH2) 2O, in the presence of NaOH....
-
Provide a mechanism for the following reaction OH H2SO4 H20
-
Provide a mechanism for the following reaction. H2SO H2O
-
Suppose you bought a new home for $210,000 using a 30-year mortgage with monthly payments of $1,218.841. The annual interest rate of the mortgage is 5.7%. After the first 3 years (36 monthly...
-
Rather than use the hypothetical information above, determine from available news sources if the Fed is increasing, holding steady, or decreasing interest rates. Similarly, is the Federal Government...
-
For a two-sided matching problem, we say that matching is Pareto efficient, if there is no matching ', such that and '(x) R (x) for all x WUF, '(x) Px (x) for some x WUF. Show that stability implies...

Study smarter with the SolutionInn App


