For each pair of compounds below, identify which compound is more acidic and explain your choice: (a)
Question:
(a)
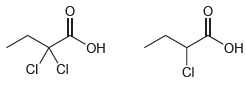
(b)
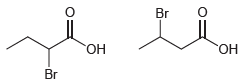
Transcribed Image Text:
ОН "он Но. ČI Br ОН он Он Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
a The compound with two chlorine atoms is more acidic because of the electron with drawi...View the full answer

Answered By

Projeet Mohanty
Growing up I had difficulties learning physics as i did not get the necessary help. Keeping that in mind I have made myself eligible enough to help people with physics. I teach students in class and make the topic interesting by providing relevant examples. Here i would like to help students with their questions and try to provide the best possible answers.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each pair of compounds below, determine whether they are identical compounds, constitutional isomers, stereoisomers, or different conformations of the same compound: (a) (b) (c) (d) (e) (f) (g)...
-
For each pair of compounds below, identify the more acidic compound: (a) (b) (c) (d) (e) (f) (g) (h) SH
-
For each pair of compounds below, identify the more acidic compound: (a) (b) (c) (d) (e) (f) (b) C (d) H. -S -3- (f) X H' `H.
-
In Exercises approximate the definite integral using the Trapezoidal Rule and Simpson's Rule with n = 4. Compare these results with the approximation of the integral using a graphing utility. 3.1 J3...
-
1) What is the best way to handle manufacturing overhead costs in order to get the most timely job cost information? A. The company should apply overhead using an estimated rate throughout the year....
-
How many joules of potential energy does a 1.5-kg book gain when it is elevated 2 m? When it is elevated 4 m? (Let g = 10 N/kg.)
-
Accounting for Participation Rates Exercises 2.30 and 2.31 show that smokers who agreed to be in the Deposit group (having their own money at risk) were much more likely to quit smoking than those...
-
Madison Services Company had the following transactions during the month of April: Apr. 2. Issued Invoice No. 201 to Triple Play Corp. for services rendered on account, $345. 3. Issued Invoice No....
-
Scot and Vidia, married taxpayers, earn $272,000 in taxable income and $9,800 in interest from an investment in City of Tampa bonds. Using the U.S. tax rate schedule for married filing jointly, how...
-
Using continuity correction, probability of getting greater than 60 successes. P(x = 60) P(x >60) P(x > 59.5) P(x > 60.5) a) b) c) d)
-
Identify the most acidic proton in each of the following compounds and explain your choice: (a) (b) 0-H F3C 0-H CI. CI
-
Consider the structure of 2,3-dichloropropanoic acid: This compound has many constitutional isomers. (a) Draw a constitutional isomer that is slightly more acidic and explain your choice. (b) Draw a...
-
Name the two types of ledgers.
-
How do B lymphocytes undergo clonal expansion and differentiation into antibody-secreting plasma cells upon encountering specific antigens, and what are the implications for long-term immunity ?
-
An investor with utility function of wealth U(w) = 0.5*ln(w) + 1 What is this investor's attitude towards risk? How does he behave when he has more wealth as opposed to when he is poor? (10 marks)
-
Discuss the phenomenon of immune tolerance, including its induction and maintenance mechanisms, and its relevance in preventing autoimmune reactions against self-antigens?
-
You trade in your old car for $5000.You can afford new car payments of $500/mo. Banks will loan at 8% interest, for 48 months.How much car can you buy?
-
A mortgage loan of $2,000,000 was made 5 years ago at 7% for a 25-year term. Now if the current market interest rate is 8%, what is the market value of the loan?
-
Use the fact that the trigonometric functions are periodic to find the exact value of each expression. Do not use a calculator. cot 17/4
-
A company has the following incomplete production budget data for the first quarter: In the previous December, ending inventory was 200 units, which was the minimum required, at 10% of projected...
-
Epoxides can be synthesized by treating halohydrins with aqueous base. Propose a mecha-nism for reactions (a) and (b), and explain why no epoxide formation is observed in (c). (a) (b) (c) CI NaOH H20...
-
Write structures for products A, B, C, and D, showing stereochemistry. (Hint: B and D are stereoisomers.) Ko Br OH (H2) TsCl, pyr OH K CO3
-
Propose mechanisms for the following reactions. (a) (b) cat. H.SO Br C3 cat. H.,SO OH
-
You observe the price of a latte changes from $4 to $5. Instructions: Round your answer to one decimal place. If you are entering a negative number include a minus sign. a. What is the percentage...
-
Layer 2 switches work at a simple level for communication and switching. With technology advancing and routers being able to make more complex decisions, why is this method still used?
-
Explore the significance of intermedial allusions in multimedia storytelling, such as in graphic novels or hypertext fiction, and their implications for narrative coherence and interpretation.

Study smarter with the SolutionInn App


