Identify the most acidic proton in each of the following compounds and explain your choice: (a) (b)
Question:
(a)
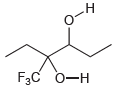
(b)
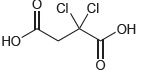
Transcribed Image Text:
0-H F3C 0-H CI. CI ОН но
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
a The highlighted proton is more acidic When this location is deprotonated the conjugate ba...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Diphenylmethane is significantly more acidic than benzene, and triphenylmethane is more acidic than either. Identify the most acidic proton in each compound, and suggest a reason for the trend in...
-
Diphenylmethane is significantly more acidic than benzene, and triphenylmethane is more acidic than either. Identify the most acidic proton in each compound, and suggest a reason for the trend in...
-
For each compound below, identify the most acidic proton in the compound: (a) (b) (c) (d) (e) (f) (g) (h) -NH2 -
-
In Exercises, analyze and sketch a graph of the function. Label any intercepts, relative extrema, points of inflection, and asymptotes. Use a graphing utility to verify your results. f(x)=x16 - x
-
Luke Skywalker College has chosen to report as a public university reporting as a special-purpose entity engaged. Deferred Revenues were reported as of July 1, 2014 in the amount of $5,000,000....
-
The following are all independent cases: Required Fill in each blank with the appropriate dollar amount. The Bike The Rental The Uniform Shop CenterCenter Sales Cost of sales Gross profit Selling...
-
A consumer advocacy agency, Equitable Ernest, is interested in providing a service that allows an individual to estimate their own credit score (a continuous measure used by banks, insurance...
-
Eastern Merchants shows the following information for its two divisions for year 1: Required Compute divisional operating income for the two divisions. Ignore taxes. How well have these...
-
A 2m rod of mass 8 kg is supported by a (zero-friction) pin-joint at A. It is released from rest from the horizontal position. When the bar reaches the position as shown in the diagram,=60. Answer...
-
Choose a case study, and then write about the ethical implications and the impact of the events that are described. Each case study includes a set of questions that you should answer. You can choose...
-
In the following compound two protons are clearly identified. Determine which of the two is more acidic. After comparing the conjugate bases, you should get stuck on the following question: Is it...
-
For each pair of compounds below, identify which compound is more acidic and explain your choice: (a)
-
Basing your answers on the general mechanism for the first stage of acid-catalyzed acetal hydrolysis
-
What is different about gauging the credit risk of a foreign buyer?
-
What is the relationship between credit and currency risk?
-
What is the rationale for hedging currency risk?
-
What is the nature of currency risk in international trade?
-
What are the major differences between the United States and Chinas balance of payments?
-
Cretin Enterprises uses a predetermined overhead rate of $7 .50 per direct labour hour. This predetermined rate was based on 16,000 estimated direct labour-hours and $120,000 of estimated total...
-
Pedro Bourbone is the founder and owner of a highly successful small business and, over the past several years, has accumulated a significant amount of personal wealth. His portfolio of stocks and...
-
Suggest an experiment using an isotopically labeled alcohol that would prove that the formation of an alkyl sulfonate does not cause cleavage at the C-O bond of the alcohol.
-
An exception to what we have just said has to do with syntheses of unsymmetrical ethers in which one alkyl group is a tert-butyl group and the other group is primary. For example, this synthesis can...
-
(a) Outline two methods for preparing isopropyl methyl ether by a Williamson ether synthesis. (b) One method gives a much better yield of the ether than the other. Explain which is the better method...
-
Give the worst-case Big O running time of this code and explain in detail how you arrived at this answer, assuming an ArrayList is passed. public static void f1( List 1st) { int i=0; while (i
-
Create a java program that will accept an integer and character. If the user input 'A', compute and display the sum of 10 integers else display "Invalid Character". run: Enter a letter: a Enter a...
-
Give the worst-case Big O running time of this code and explain in detail how you arrived at this answer. for(int j = 0; j < numItems; j++) { int i = numItems; while (i > 0) = i i 3; // integer...

Study smarter with the SolutionInn App


