For each pair of compounds, determine which C = C bond will produce a stronger signal. a.
Question:
a.
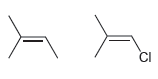
b.
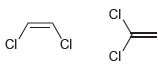
Transcribed Image Text:
CI CI CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a The presence of the Cl will cause the CC bond ...View the full answer

Answered By

Hamza Amjad
Currently I am student in master degree program.from last two year I am tutring in Academy and I tought many O/A level student in home tution.
4.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each pair of compounds, circle the compound you expect to have the higher boiling point. Explain your reasoning. (a) (CH3)3C-C(CH3)3 and (CH3)2CH-CH2CH2-CH(CH3)2 (b) CH3(CH2)6CH3 and...
-
For each pair of compounds, predict the one with a higher boiling point. Which compounds have zero dipole moments? (a) cis-1,2-dichloroethene or cis-1,2-dibromoethene (b) cis- or...
-
For each pair of compounds, describe which instrumental technique (IR, MS, proton NMR, carbon NMR) you could use to distinguish for certain which of the two compounds was in a sample. Describe what...
-
The annual revenues associated with several large apartment complexes are $300, $450, $425, $50, $75, and $150 for years 0, 1, 2, 3, 4, and 5, respectively. Determine the net cash flow and whether...
-
Suppose T1(N) = O(f (N)) and T2(N) = O(f (N)). Which of the following are true? a. T1(N) + T2(N) = O(f (N)) b. T1(N) T2(N) = o(f (N)) c. T1(N) / T2(N) = O(1) d. T1(N) = O(T2(N))
-
Aspartame is an artificial sweetener that is 160 times sweeter than sucrose (table sugar) when dissolved in water. It is marketed as NutraSweet. The molecular formula of aspartame is C 14 H 18 N 2 O...
-
A process for the manufacture of film has performed in the past with an average of 0.8 imperfections per 10 linear feet. (a) Construct a chart to be used in the inspection of 10 -foot sections. (b)...
-
The income statement and additional data of Breen Travel Products, Inc., follow: Additional data: a. Acquisition of plant assets was $200,000. Of this amount, $160,000 was paid in cash and $40,000 by...
-
Circular curves chords It is required to truncate a corner by 3 equal chords 20m long. The three-chord truncation is required to be calculated and set-out from the intersection point (IP). Determine...
-
The position of a particle as a function of time is given by r(vector) = (5.0i + 4.0j)t 2 m, where t is in seconds. a. What is the particles distance from the origin at t = 0, 2, and 5 s? b. Find an...
-
Because there are _______, adjusted gross income is always ______ taxable income. a. Deductions and exemptions; less than b. Deductions and exemptions; greater than c. Credits; greater than d....
-
The alternative minimum tax has the effect of limiting a. Income. b. Taxable income. c. Deductions. d. Exemptions.
-
In alphabetical order below are current asset items for Wallace Companys balance sheet at December 31, 2012. Prepare the current assets section (including a complete heading). Accounts receivable...
-
Identify a source and a control document that Chris Condie Computer Supplies could use for each of the following transactions: Transaction Repairs a customer's computer on credit Transfers monthly...
-
C Craft has been having trouble collecting monies owed from accounts receivable and has decided to apply the interest rates that are shown on the customers tax invoices for amounts that are overdue....
-
Indicate the journal C Namble would use to enter the following transactions. Transaction C Namble remitted wages C Namble received a cheque from F Lee C Namble purchased goods on credit from P Way C...
-
R Pessotto sold a truck (motor vehicle) to J Aitkens for $5720 ($5200 + $520 GST) who paid with a direct deposit into the bank account. Prepare the journal for R Pessotto.
-
Matthews paid $3850 ($3500 + $350 GST) to R Badger for an office desk, cabinet and chair (office furniture). Prepare the journal for S Matthews.
-
Develop a list of the major stakeholders of your college or university. What priority does the school assign to these stakeholders ? Do you agree or disagree with this priority? Explain your...
-
a. Determine the domain and range of the following functions.b. Graph each function using a graphing utility. Be sure to experiment with the window and orientation to give the best perspective of the...
-
How would you synthesize the following substances starting frombenzene? ( C-CH2 .CH- (a) (c) H
-
The compound MON-0585 is a nontoxic, biodegradable larvicide that is highly selective against mosquito larvae. Synthesize MON-0585 using either benzene or phenol as a source of the aromaticrings....
-
Hexachlorophene, a substance used in the manufacture of germicidal soaps, is prepared by reaction of 2, 4, 5-trichiorophenol with formaldehyde in the presence of concentrated sulfuric acid. Propose a...
-
The initial margin on a GBP futures contract is $2035 and maintenance is $1850. You buy one contract (62,500 Pounds) at $1.3100 and place $2035 in your account. The price of your contract drops to...
-
1.Define a).Market research transnational firm b).Target market c).Maslow's hierarchy of needs d).ethnocentric polycentric Geocentric Explain Please create a response to the question below that is...
-
Describe one product development recommendation and one market development recommendation for Tesla. Explain why this idea should be adopted by the Tesla EV company in the New Zealand market.

Study smarter with the SolutionInn App


