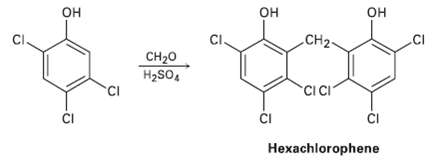
Hexachlorophene, a substance used in the manufacture of germicidal soaps, is prepared by reaction of 2, 4,
Question:
Hexachlorophene, a substance used in the manufacture of germicidal soaps, is prepared by reaction of 2, 4, 5-trichiorophenol with formaldehyde in the presence of concentrated sulfuric acid. Propose a mechanism for thereaction.
Transcribed Image Text:
н он он CI Он CI. CH2- сн20 H2SO, CIC CI ČI ČI Hexachlorophene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
DR1 Formaldehyde is protonated to form a carbocation 2 0 H CI OH H CHOH OH Base OH The formal...View the full answer

Answered By

Amit Kumar
I am a student at IIT Kanpur , which is one of the prestigious colleges in INDIA.
Cleared JEE Advance in 2017.I am a flexible teacher because I understand that all students learn in different ways and at different paces. When teaching, I make sure that every student has a grasp of the subject before moving on.
I will help student to get the basic understanding clear. I believe friendly behavior with student can help both the student and the teacher.
I love science and my students do the same.
4.90+
44+ Reviews
166+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following reaction is important in the manufacture of sulfuric acid. At 900 K, 0.0216 mol of SO2 and 0.0148 mol of O2 are sealed in a 1.00-L reaction vessel. When equilibrium is reached, the...
-
A press produces parts used in the manufacture of large-screen plasma televisions. If the press is correctly adjusted, it produces parts with a scrap rate of 5%. If it is not adjusted correctly, it...
-
The strength of paper used in the manufacture of cardboard boxes (y) is related to the percentage of hardwood concentration in the original pulp (x). Under controlled conditions, a pilot plant...
-
R and Q form equal partnership RQ on February 15. Partner R has a tax year ending on July 31, whereas partner Qs tax year ends on October 31. What taxable year must the partnership use? a. February...
-
Determine the gross income of the beneficiaries in the following cases: a. Justin's employer was downsizing and offered employees an amount equal to one year's salary if the employee would...
-
Starting with benzene and any reagents with three or fewer carbon atoms, show how you would prepare each of the following compounds: (a) (b) (c) (d) Z NH2 CI .CI I
-
The Wind Map The website hint.fm/wind/ shows the current wind patterns across the US. In order to generate this map, what two variables are being recorded at weather stations across the US?
-
Horton Manufacturing Inc. produces blinds and other window treatments for residential homes and offices. The owner is concerned about the maintenance costs for the production machinery, as...
-
Coolbrook Company has the following information available for the past year: sales revenue Cost of goods sold and operating expenses Net operating income Average invested assets The company's hurdle...
-
The bank portion of the bank reconciliation for Phillips Company at October 31, 2014, was as follows. The adjusted cash balance per bank agreed with the cash balance per books at October 31. The...
-
The compound MON-0585 is a nontoxic, biodegradable larvicide that is highly selective against mosquito larvae. Synthesize MON-0585 using either benzene or phenol as a source of the aromaticrings....
-
Benzenediazonium carboxylate decomposes when heated to yield N2, C02, and a reactive substance that can't be isolated. When Benzenediazonium carboxylate is heated in the presence of furan, the...
-
Determine the force F at the end of the beam C so that the displacement at C is zero. EI is constant. Use the moment-area theorems. B-
-
What strategy/tactic might marketers use to see which competitors' items are competing against their own items and to identify market segments so they can gauge how well their items are positioned to...
-
1. Determine the term "culture." Describe one analytical framework that may be used to measure and analyze the culture of a society. 2. Religion is an element of culture. How does the religion of...
-
Choose a recent television commercial and identify the objectives each appears to attempt to accomplish. Inquiry: If you were a marketer working for this company, what procedure would you use to...
-
Do you think organizations that promote professional ethics are effective? Why or why not? 2. What should employees do when one of their colleagues or a company executive is being investi-gated for...
-
When do you use your mental shortcut (system 1) to make purchase decisions?When do you use deliberate and rational thinking (system 2) to make purchase decisions? What are the pros and cons of...
-
The city of Dystopia experienced difficulties with groups of rowdy individuals congregating on downtown sidewalks and harassing passersby. When the local police were unable to control the situation...
-
On August 31, 2012, the balances of the accounts appearing in the ledger of Wood Interiors Company, a furniture wholesaler, are as follows:Prepare the August 31, 2012, closing entries for Wood...
-
N-butane is to be liquefied to make liquid petroleum gas (LPG). The butane is available at 25 C and 1 bar, it will be compressed to 15 bar in a compressor that has an isentropic efficiency of 85%,...
-
The oxymercuration reaction can be run in a methanol as the solvent rather than water. Predict the product of this reaction. 1) Hg(O,CCH)2, CH3OH 2) NaBH4, NAOH CH,CH,CH CH=CH;
-
The tautomerization of an enol to a ketone is catalyzed by either acid or base. In the acid-catalyzed mechanism H+ is added in the first step (see Figure). In the base-catalyzed mechanism, H+ is...
-
An unknown compound has the formula C6H10. (a) What is the DU for this compound? (b) When a solution of Br2 in CC14 is added to the unknown, the bromine color disappears. What information does this...
-
Aaron, Deanne, and Keon formed the Blue Bell General Partnership at the beginning of the current year. Aaron and Deanne each contributed $138,000, and Keon transferred an acre of undeveloped land to...
-
The following information pertains to the inventory of Parvin Company: Jan. 1 Apr. 1 Oct. 1 Beginning inventory Purchased Purchased 400 units @ 2,400 units @ 1,100 units $17 $22 $23 During the year,...
-
Gold Nest Company of Guandong, China, makes birdcages for the South China market. The company sells its birdcages through an extensive network of street vendors who receive commissions on their...

Study smarter with the SolutionInn App


