For the substitution reaction shown below, assume a stepwise process is taking place and draw the mechanism.
Question:
For the substitution reaction shown below, assume a stepwise process is taking place and draw the mechanism.
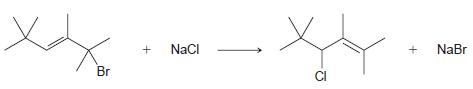
Transcribed Image Text:
+ NaCI NaBr Br CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Br...View the full answer

Answered By

Gauri Hendre
I worked as EI educator for Eduphy India YT channel. I gave online tutorials to the students who were living in the villages and wanted to study much more and were preparing for NEET, TET. I gave tutions for topics in Biotechnology. I am currently working as a tutor on course hero for the biochemistry, microbiology, biology, cell biology, genetics subjects. I worked as a project intern in BAIF where did analysis on diseases mainly genetic disorders in the bovine. I worked as a trainee in serum institute of India and Vasantdada sugar institute. I am working as a writer on Quora partner program from 2019. I writing on the topics on social health issues including current COVID-19 pandemic, different concepts in science discipline. I learned foreign languages such as german and french upto A1 level. I attended different conferences in the science discipline and did trainings in cognitive skills and personality development skills from Lila Poonawalla foundation. I have been the member of Lila poonawalla foundation since 2017. Even I acquired the skills like Excel spreadsheet, MS Office, MS Powerpoint and Data entry.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) What is the difference between a substitution reaction and an addition reaction? Which one is commonly observed with alkenes and which one with aromatic hydrocarbons? (b) Using condensed...
-
The rate law for the substitution reaction of 2-bromobutane and HO- in 75% ethanol and 25% water at 30C is rate = 3.20 10-5[2-bromobutane][HO-] + 1.5 10-6[2-bromobutane] What percentage of the...
-
Show how you might use a nucleophilic substitution reaction of 1-bromopropane to synthesize each of the following compounds. (You may use any other compounds that are necessary.) (a) (b)...
-
The table lists the actual annual cost y to drive a midsize car 15,000 miles per year for selected years X. (a) Predict whether the correlation coefficient is positive, negative, or zero. (b) Find a...
-
Here is the "Scenario" Online Ordering and Delivery Service for Sandwich Shop Joe's Sub Shop is starting an online ordering system that will provide local delivery within five miles of the sub shop....
-
What are the various types of chain drive? Explain with neat sketches.
-
What is meant by dual dating an audit report?
-
Zhu Manufacturing is considering the introduction of a family of new products. Long- term demand for the product group is somewhat predictable, so the manufacturer must be concerned with the risk of...
-
Tigreal Inc. purchases a patent from Leonil Company for $28,000 in cash. The patent has seven years remaining in its term. What is the entry to record the purchase?
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
When a nucleophile and electrophile are tethered to each other (that is, both present in the same compound), an intramolecular substitution reaction can occur, as shown. Assume that this reaction...
-
The reaction below exhibits a second-order rate equation: (a) What happens to the rate if the concentration of 1-iodopropane is tripled and the concentration of sodium hydroxide remains the same? (b)...
-
Do you think most employees know what the mission of their company is? Suggest some ways leaders can effectively communicate the mission to people both inside and outside the organization.
-
Troy Engines, Limited, manufactures a variety of engines for use in heavy equipment. The company has always produced all of the necessary parts for its engines, including all of the carburetors. An...
-
Write a recursive function printFlag(A) that takes an array of integers as input, prints a flag-shape numbers from it such that the first level has all array elements. From then, at each level the...
-
What is the output of the following #include ?code using namespace std; int fun(int a, int b = 1, int c=2) { } retum (a+b+c); int main() { cout < < fun(12,,2); } retum 0;
-
What is the output of the ?following code #include using namespace std; int xyz (int x, int y) { int result; result = 0; { while (y != 0) result = result + x;B y = y 1; } return(result); } int main...
-
What is the output of the #include using namespace std; ?following code int main() { int num[] {6, 5, 7, 3, 6, 7, 3, 3, 3, 9, 6, 7}; int a = 0; for(int i=0; i < 5; i++){ for (int j=0; j < 12; j++){...
-
Use Exercise 22 to find the centroid of a quarter-circular region of radius a.
-
Research an article from an online source, such as The Economist, Wall Street Journal, Journal of Economic Perspectives, American Journal of Agricultural Economics, or another academic journal. The...
-
Predict the outcome of the following reactions: (a) (b) CN CI 2 equiv. KNH2 liq. NH3 -33 C NH CH3 2 equiv. NaNH liq. NH2 -33C Cl
-
Your laboratory instructor gives you a mixture of 4-methylphenol, benzoic acid, and toluene. Assume that you have available common laboratory acids, bases, and solvents and explain how you would...
-
Explain how it is possible for 2, 29-dihydroxy-1, 19-binaphthyl (shown at right) to exist in enantiomeric forms. OH
-
Managers use CVP concepts to perform sensitivity analysis. Sensitivity analysis is a "what-if" technique that asks what will happen to a company's breakeven or target profit if sales price, costs, or...
-
John Fleming, chief administrator for Valley View Hospital, is concerned about the costs for tests in the hospital s lab. Charges for lab tests are consistently higher at Valley View than at other...
-
A company is looking at new equipment with an installed cost of $436,859. This cost will be depreciated straight-line to zero over the project's 6-year life, at the end of which the equipment can be...

Study smarter with the SolutionInn App


