How many signals would you expect in the 1 H NMR spectrum of each of the following
Question:
(a)
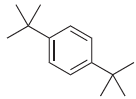
(b)
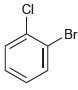
(c)
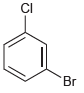
(d)
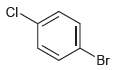
(e)
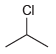
(f)
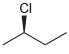
Transcribed Image Text:
CI Br ū-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
a 2 ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A compound with molecular formula C 17 H 36 exhibits a 1 H NMR spectrum with only one signal. How many signals would you expect in the 13C NMR spectrum of this compound?
-
How many signals would you expect in the 13 C NMR spectrum of each of the compounds in Problem 16.34? In Problem 16.34 How many signals would you expect in the 1 H NMR spectrum of each of the...
-
A compound with molecular formula C 8 H 18 exhibits a 1 H NMR spectrum with only one signal. How many signals would you expect in the 13 C NMR spectrum of this compound?
-
A cheetah spots a Thomsons gazelle, its preferred prey, and leaps into action, quickly accelerating to its top speed of 30 m/s, the highest of any land animal. However, a cheetah can maintain this...
-
1. What is the function of Apple's iBeacon? 2. What are the differences between iBeacon and GPS? 3. How might a retail business benefit from iBeacon? 4. What are some of the concerns that customers...
-
In 1865, Jules Verne wrote a story in which three men went to the Moon by means of a shell shot from a large cannon sunk in the ground. (a) What muzzle speed must the cannon have in order for the...
-
A flywheel, with a mass of \(50 \mathrm{~kg}\) and an eccentricity of \(12 \mathrm{~mm}\), is mounted at the center of a steel shaft of diameter \(25 \mathrm{~mm}\). If the length of the shaft...
-
Assume Ashland Community Hospital had the following supplies costs for two products used in its operating room. Standard costs for one surgery: Item X, 5 pieces at $50 each; Item Y, 10 pieces at $75...
-
Walnut, Incorporated has received a special order for 2,000 units of its product at a special price of $150. The product normally sells for $200 and has the following manufacturing costs: Direct...
-
Mr B aged 52 years, has earned rupees 75,00,000 out of his business. His ex-wife gifted him a car worth rupees 8 lakh. He spent a total of rupees 20 lakh during a family trip. He won a lottery of 16...
-
Because of crop failures last year, the San Joaquin Packing Company has no funds available to finance its canning operations during the next six months. It estimates that it will require $1,200,000...
-
How would you distinguish between the following compounds using 13 C NMR spectroscopy?
-
The following facts pertain to a noncancelable lease agreement between Alschuler Leasing Company and McKee Electronics, a lessee, for a computer system. Inception date ........... October 1, 2014...
-
Sain Corp. has preferred stock that was issued 3 years ago. It currently offers a return of 7.2% and pays a dividend of $2.25 per share. What is the price on the preferred stock?
-
Using the 2020/21 threshold, if Bob earns gross wages of $1000/week, what effect, if any, would having a HELP Debt have on his net pay, and why? (reference about HELP...
-
You are trying to estimate the risk free rate for Peru. Suppose you don't trust the yields on Peruvian C-bonds and want to use another approach which relies on the US risk free rate and scales it up...
-
The Peru-issued USD Bond has a maturity date of 1/23/2031 and offers a coupon of 2.7830%. The payment of the coupon will take place 2.0 times per biannual on the 23.07. At the current price of...
-
Use the currency converter. In your answers, record only the numbers in bold text. Do not round up or down. For example, if the answer is 46.79 552, then record only 46.79. 1 U.S. Dollar =...
-
Evaluate the expression. -|-12|
-
The May 2014 revenue and cost information for Houston Outfitters, Inc. follow: Sales Revenue (at standard).............. $ 540,000 Cost of Goods Sold (at standard) ..........341,000 Direct Materials...
-
The Ka for dichloroacetic acid is 3.32 x 102. Approximately what percentage of the acid is dissociated in a 0.10M aqueous solution?
-
Calculate the percentage of dissociated and un-dissociated forms present in the following solutions: (a) 0.0010M glycolic acid (HOCH2CO2H; pKa = 3.83) at ph = 4.50 (b) 0.0020M propanoic acid (pKa =...
-
Which would you expect to be a stronger acid, the lactic acid found in tired muscles or acetic acid? Explain. Lactic acid CH
-
Blue-Eyed Beauty Supply is the talk of the town. When Elaine started her business, she had no idea it would evolve to be the number one supplier of cosmetology supply in the region. While her...
-
Mystic Lake Inc. bottles and distributes spring water. On July 9 of the current year, Mystic Lake reacquired 5,700 shares of its common stock at $78 per share. On September 22, Mystic Lake sold 4,000...
-
1. Find the electric flux emanating from a point charge of 6 micro coulombs. 2.An electric field of 500 V/m makes an angle of 30.00 with the surface vector, which has a magnitude of 0.500 m 2 . Find...

Study smarter with the SolutionInn App


