Which would you expect to be a stronger acid, the lactic acid found in tired muscles or
Question:
Which would you expect to be a stronger acid, the lactic acid found in tired muscles or acetic acid? Explain.
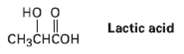
Transcribed Image Text:
но о Lactic acid CHзснҫон
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
You would expect lactic acid t...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which compound would you expect to be a stronger acid? Why? CH C-O-H or CH,S -O-H
-
Which would you expect to be the more ductile element, (a) Ag or Mo, (b) Zn or Si? In each case explain your reasoning.
-
Which alkyl halide would you expect to be more reactive in an E2 reaction? a. b. c. d. CH3 CHCHCHCH.CH, or CH.CHCH CHCH. Br Br Br CH3CH2CH CHCH3 or CH3CH2CHCH CH3 CH2CHCH2CH3 or CH2CH CHCH3
-
STEELCO manufactures two types of steel (steel 1 and steel 2) at two locations (plant 1 and plant 2). Three resources are needed to manufacture a ton of steel: iron, coal, and blast furnace time. The...
-
1. How did the differences between Japan's and the United States' national cultural values affect the interaction between Norio and Michael? Use the information on PPT 8-3 and 8-4 to support your...
-
Mackin Investing charges its customers a 1% commission. The Ross Group, a discount broker, charges $25 per trade. For what amount of stock would both brokers charge the same commission?
-
How do you define, describe, and represent multi-level modeling constructs or template for modeling Mission System and Enabling System operations. What is the purpose of the system behavioral...
-
Butler Corporation has three divisions, each operating as a responsibility center. To provide an incentive for divisional executive officers, the company gives divisional management a bonus equal to...
-
Pizza Corporation acquired 75 percent of Slice Corporation's voting common stock on January 1, 20x4, for $351,000, when the fair value of its net identifiable assets was $464,000 and the fair value...
-
Claytonhill Beverages Ltd. is 100 percent owned by Buzz Bottling. While the company has in the past been profitable, it incurred a loss for the year ended December 31, 2012. The parent company, Buzz...
-
Calculate the percentage of dissociated and un-dissociated forms present in the following solutions: (a) 0.0010M glycolic acid (HOCH2CO2H; pKa = 3.83) at ph = 4.50 (b) 0.0020M propanoic acid (pKa =...
-
Dicarboxylic acids have two dissociation constants, one for the initial dissociation into a monoanion and one for the second dissociation into a dianion. For oxalic acid, HO2CCO2H, the first...
-
The following time series graph shows daily closing stock prices (adjusted for splits and dividends) for Google Inc. from January 1, 2008, through June 21, 2013. a) Which time series components are...
-
There are several pitfalls and common misconceptions in accounting analysis that an analyst should avoid. Discuss these pitfalls and misconceptions.
-
The value of assets amounts to 1,700,000 , the expected instantaneous rate of return and corresponding volatility are 8 and 13 percent. Time to maturity of the outstanding debt is 1 year. Investors...
-
The Sarbanes Oxley Act (SOX) was passed after the Enron scandal in 2002. Please address the 1 and 2 below. 1. is the role of SOX in policing corporate ethics? Do you believe (SOX) it is effective?...
-
Annual interest rates are 10%. You buy a smartphone for $300 and use it for two years. Each month, you pay a $30 fee for your asm data plan. At the end of two years, you decide to upgrade to the...
-
he treasurer at Dell estimated the expected return on equity for the firm is 14.79%. The current T-bill rate is 5.90%, the expected market return is 16.61% and the market premium is 10.71%. What is...
-
7.6 We have stressed that the wavefunction of a quantum mechanical particle does not directly represent the trajectory of that particle through space. This is very unlike the way in which we...
-
The registrar of a college with a population of N = 4,000 full-time students is asked by the president to conduct a survey to measure satisfaction with the quality of life on campus. The following...
-
Calculate the fraction of empty space in cubic closest packing to five significant figures.
-
Explain how it is possible for 2, 29-dihydroxy-1, 19-binaphthyl (shown at right) to exist in enantiomeric forms. OH
-
Phenols are often effective antioxidants (Problem 21.26 and "The Chemistry of . . . Antioxidants" in Section 10.11) because they are said to "trap" radicals. The trapping occurs when phenols react...
-
A compound X (C10H14O) dissolves in aqueous sodium hydroxide but is insoluble in aqueous sodium bicarbonate. Compound X reacts with bromine in water to yield a dibromo derivative, C10H12Br2O. The...
-
The setting of a piece of literature may be an important factor that influences the actions of the characters. Two literary works in which the setting plays an important role are The Story of an Hour...
-
Water is flowing in the pipe shown in the figure below, with the 8.45-cm diameter at point 1 tapering to 3.60 cm at point 2, located y = 13.0 cm below point 1. (a) point 1 m/s (b) point 2 m/s
-
A certain target is bombarded by electrons in an x-ray tube. The K, L, and M energy levels have energies 84.7, 14.3, and 2.97 keV, respectively. (a) What is the minimum value of the accelerating...

Study smarter with the SolutionInn App


