How would you use IR spectroscopy to distinguish between the following pairs of compounds? (a) (b) N.
Question:
(a)
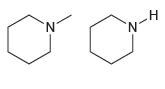
(b)
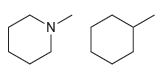
Transcribed Image Text:
N.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
a The second compound will have an NH stretching signal between 3300 ...View the full answer

Answered By

Sandip Agarwal
I have an experience of over 4 years in tutoring. I have solved more than 2100 assignments and I am comfortable with all levels of writing and referencing.
4.70+
19+ Reviews
29+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How might you use IR spectroscopy to distinguish among the three isomers 1 -butyne, 1, 3-hutadiene, arid 2-butyne?
-
How could you use IR spectroscopy to distinguish between the isomers 1-hexyne and 1,3-hexadiene?
-
How could you use IR spectroscopy to distinguish between the following pairs of isomers? a. b. c. (CH3CH2)3N and (CH3CH2CH2)2NH 0 CH.CCH,CH, and CH CHCH-CH, CH3 CHCHO and CH CHOCH
-
Define variable. Also discuss variable initialization.
-
The complexity of interpersonal relationships increases dramatically as the size of a group increases. Determine the numbers of different two-person relationships in groups of people of sizes (a) 3,...
-
Although everyone makes mistakes, describe how history repeats itself when there are patterns of progress and regression in the provision of patient care. In forming your answer, consider how...
-
Determine the torsional strain energy stored in the tapered rod when it is subjected to the torque \(\mathbf{T}\). The rod is made of material having a modulus of rigidity of \(G\). L 2ro To T
-
The following unadjusted trial balance is prepared at fiscal year-end for Nelson Company. Rent expense and salaries expense are equally divided between selling activities and the general and...
-
Elizabeth and Robert are a young couple who recently got married. They wish to have children, but before trying to conceive, they have genetic testing done and find out that they are both carriers of...
-
Consider the following 0x86 program: .data array DWORD 1,2,3,4,5,6,7,8,9 aravSize -(S-array)/4 ; array .code main PROC mov ecx,arraySize-1 mov esi.OFFSET array L1: mov eax. [esi] sda mov bx,2 idiv bx...
-
A compound with molecular formula C 5 H 13 N exhibits three signals in its proton NMR spectrum and no signals above 3000 cm 1 in its IR spectrum. Draw two possible structures for this compound.
-
Discuss opportunities and risks associated with each brothers proposal.
-
Calculate the electronic gas pressure in metallic sodium, at T = 0, in which the concentration of free electrons is n = 2.5.1023 cm -3. Use the equation for the pressure of ideal gas.
-
f(x) = = 3 1++ dt. What is (f) '(0)?
-
For each of the functions given below, find the least positive integer n such that f(x) is O(n) for each of the following functions: (a) f(x)=3+5* (b) f(x)=3.5 (c) f(x) = 2 log(2) (d) f(x) = 7 x7
-
Evaluate the indefinite integral. 2x + 5 -dx = x+1 +C.
-
At the end of the current fiscal year, ABC Corporation had the following balances: Assets $50,000 Common Stock $30,000 Dividends Declared $5,000 Expenses $75,000 Liabilities $20,000 Revenues $90,000...
-
A leased a building to B for a ten-year term at an annual rental of 200,000. At the inception of the lease, A received 800,000, which covered the first two years of rent of 400,000 and a security...
-
Show that the product of the roots of a quadratic equation is c/a.
-
A 20-cm-square vertical plate is heated to a temperature of 30oC and submerged in glycerin at 10oC. Calculate the heat lost from both sides of the plate.
-
For each compound, predict the major product of free-radical bromination. Remember that bromination is highly selective, and only the most stable radical will be formed. (a) Cyclo-hexane (b)...
-
When exactly 1 mole of methane is mixed with exactly 1 mole of chlorine and light is shone on the mixture, a chlorination reaction occurs. The products are found to contain substantial amounts of...
-
The chlorination of pentane gives a mixture of three mono-chlorinated products. (a) Draw their structures. (b) Predict the ratios in which these mono-chlorination products will be formed, remembering...
-
Calculating Present Values Imprudential, Inc., has an unfunded pension liability of $645 million that must be paid in 25 years. To assess the value of the firm's stock, financial analysts want to...
-
Describe the operating activities of each company noting similarities and difference between COCA COLA & PEPSICO. Identify two economy wide factors and industry wide factors that could impact on the...
-
The trial balance for a company listed the following account balances at December 31, Year 1, the end of its fiscal year: cash, $36,000; accounts receivable. $31,000; Inventory, $45,000; equipment...

Study smarter with the SolutionInn App


