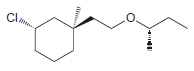
Identify the configuration of each chirality center in the following compounds: a. b. c. d. e. f.
Question:
a.
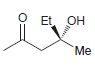
b.
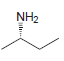
c.
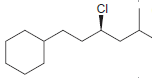
d.
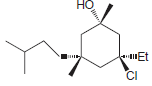
e.
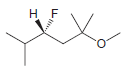
f.
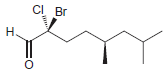
g.
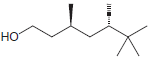
h.
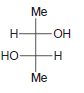
i.
Transcribed Image Text:
Et OH Me NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
a b c ...View the full answer

Answered By

Antony Sang
I am a research and academic writer whose work is outstanding. I always have my customer's interests at heart. Time is an important factor in our day to day life so I am always time conscious. Plagiarism has never been my thing whatsoever. I give best Research Papers, Computer science and IT papers, Lab reports, Law, programming, Term papers, English and literature, History, Math, Accounting, Business Studies, Finance, Economics, Business Management, Chemistry, Biology, Physics, Anthropology, Sociology, Psychology, Nutrition, Creative Writing, Health Care, Nursing, and Articles.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Assign the configuration of each chirality center in the following compounds: (a) (b) (c) (d) (e) C=0 - - - H,OH %3D - - - H,OH
-
Seventeen of the 20 naturally occurring amino acids (See the following table) exhibit exactly one chirality center. Of the remaining three amino acids, glycine has no chirality center, and the other...
-
Each of the following compounds possesses carbon atoms that are chirality centers. Locate each of these chirality centers, and identify the configuration of each one: a. b. c. d. e. f. Ephedrine A...
-
A Contractors Ltd was formed on 1 January 2012 and the following purchases and sales of machinery were made during the first 3 years of operations. Each machine was estimated to last 10 years and to...
-
What recommendations would you make to Howard Schultz to sustain the company's growth and support continued strong financial performance in the years ahead?
-
Using seasonal relatives. Apples Citrus Fruit Farm ships boxed fruit to anywhere in the world. Using the following information, a manager wants to forecast shipments for the first four months of next...
-
Assume you prepay Hercules Workout World for a package of six physical training sessions. Which type of account should you have in your records? a. Prepaid expense b. Accrued expense c. Accrued...
-
Enders, Inc., a domestic corporation that invests in foreign securities, reports total taxable income for the tax year of $290,000, consisting of $208,800 in U.S.-source business profits and $81,200...
-
At the end of 2019, Alonzo Company's manufacturing inventory and expense accounts held the following costs. Work in Process Inventory Direct Materials P120.000 Direct Labor Factory Overhead P60.000...
-
Two homogeneous dielectric regions 1 (p < 4 cm) and 2 (p > 4 cm) have dielectric constants 3.5 and 1.5, respectively. If D2 = 12ap - 6a0 + 9az nC/m2, calculate: (a) E1 and D1, (b) P2 and ppv2, (c)...
-
For each pair of compounds, identify which compound is more acidic and explain your choice. (a) 2,4-Dimethyl-3,5-heptanedione or 4,4-Dimethyl-3,5-heptanedione (b) 1,2-Cyclopentanedione or...
-
In Chapter 10, we will see that an acetylide ion (formed by treatment of acetylene with a strong base) can serve as a nucleophile in an S N 2 reaction: This reaction provides a useful method for...
-
Indicate a good method for evaluating the integral (but do not evaluate). Your choices are substitution (specify u and du), Integration by Parts (specify u and dv), a trigonometric method, or...
-
Find the inductive reactance per mile of a single-phase overhead transmission line operating at 60 Hz, given the conductors to be Partridge and the spacing between centers to be 20 ft.
-
A laser beam has a radius of \(1 \mathrm{~mm}\). How powerful does the laser have to be for the maximum magnitude of the magnetic field in the beam to be \(4 \mu \mathrm{T}\) ?
-
A new fractal surface is being developed for heat transfer by Crinkle, Inc. It is projected to revolutionize the home heating industry. As an agent of Industrial Espionage Ltd., you have stolen some...
-
Use the national income identity GDP = C + I + G + X to explain what a current account deficit (negative net exports) means in terms of domestic spending, production, and borrowing.
-
Consider a bank of three single-phase two-winding transformers whose high-voltage terminals are connected to a three-phase, 13.8 k V 13.8 k V feeder. The low-voltage terminals are connected to a...
-
The assurance standards do not contain a requirement that auditors obtain a. Adequate knowledge in the subject matter of the assertions being examined. b. An understanding of the auditees internal...
-
Two mutually exclusive investment alternatives are being considered. Alternative A requires an initial investment of $20,000 in a machine. Annual operating and maintenance costs are anticipated to be...
-
Predict the major product that is obtained when the following alkyl halides is treated with potassium terf-butoxide. Explain your reasoning. OCH CH CHI
-
Use a hybridization argument to predict the geometry of The neutral compound Pd(PPh3)4.
-
The Wilkinson catalyst chlorotris (triphenylphosphine) rhodium(I), ClRh(PPh0)3, brings about the catalytic hydrogenation of an alkene in homogeneous solution: (a) Using the following mechanistic...
-
The Cited paper is about Sustainability in Organizations: A case of Public companies in Nigeria. What are the prevailing themes that link the academic articles? (What is the common topic matter among...
-
1. This week covers IT Project Management. History has shown many projects fail to deliver within budget or do not meet customer expectations. The text does not address this item in any great deal...
-
According to the Josephine Institute, rank the core ethical values in order. After ranking them, discuss how you would apply them when faced with a situation where you must choose between right and...

Study smarter with the SolutionInn App


