Identify the hydroxyaldehyde that will cyclize under acidic conditions to give the following hemiacetal: -OH
Question:
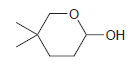
Transcribed Image Text:
-OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)

Answered By

RADHIKA MEENAKAR
I am a qualified indian Company Secretary along with Masters in finance with over 6 plus years of professional experience. Apart from this i am a certified accounts and finance tutor on many online platforms.
My Linkedin profile link is here https://www.linkedin.com/in/radhika-meenakar-88b9808a/
5.00+
12+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When ethylene oxide is treated with a strong nucleophile, the epoxide ring is opened to form an alkoxide ion that can function as a nucleophile to attack another molecule of ethylene oxide. This...
-
The mechanism for acidic hydrolysis of a nitrile resembles the basic hydrolysis, except that the nitrile is first protonated, activating it toward attack by a weak nucleophile (water). Under acidic...
-
Consider the three constitutional isomers of dioxane (C 4 H 8 O 2 ): One of these constitutional isomers is stable under basic conditions as well as mildly acidic conditions and is therefore used as...
-
Krell Industries has a share price of $22 46 today. If Krell is expected to pay a dividend of S0.83 this year, and its stock price is expected to grow to $24.11 at the end of the year, what is...
-
Eason Company manufactures wheel rims. The controller expects the following ABC allocation rates for 2018: Eason produces two wheel rim models: standard and deluxe. Expected data for 2018 are as...
-
You have a collection of \(1.0 \mathrm{k} \Omega\) resistors. How can you connect four of them to produce an equivalent resistance of \(0.25 \mathrm{k} \Omega\) ?
-
For the data in Exercise 7: a. Compute the expected frequencies for testing H0 : p1 = 0.4, p2 = 0.3, p3 = 0.1, p4 = 0.15, p5 = 0.05. b. Is it appropriate to perform a chi-square test for the...
-
Kaizer Plastics produces a variety of plastic items for packaging and distribution. One item, container #145, has had a low contribution to profits. Last year, 20,000 units of container #145 were...
-
Matrix equation to two simultaneous linear equations and working please -1 2 x Write the matrix equation = as a system of two simultaneous linear equations in -2 3 x and y. Equation 1: Equation 2:
-
1. Evaluate Nikes response to societal and consumer concerns about its contract manufacturing. 2. What are the challenges facing Nike in the future? Phil Knight and his University of Oregon track...
-
Draw the cyclic hemiacetal that is formed when each of the following bifunctional compounds is treated with aqueous acid: (a) (b) (c) (d) H.
-
The following compound has one aldehyde group and two OH groups: Under acidic conditions, either one of the OH groups can function as a nucleophile and attack the carbonyl group, giving rise to two...
-
What is white space, and why is it important?
-
Find the real solutions of the equation. X 2+x-2=0
-
National University MGT608 What do you think will be some of the most important innovations in logistics over the next ten years? As you identify these large innovations, discuss how they will affect...
-
What are 2 elements from macro and micro environment that affect under armours overall marketing strategy? segmentation approach for under armour ? what is under armors target audience or markets?...
-
The use of confirmations for cash balances and trade accounts receivable is considered a generally accepted auditing standard. However, auditors may decide not to use them if suitable alternative...
-
d. (FILL IN THE TABLE BELOW) Calculate the APPROXIMATE new price of the bond using your measure of modified duration and the percentage change in the price of the bond for the following interest...
-
Use the data set to (a) Find the sample mean, (b) Find the sample standard deviation, and (c) Construct a 99% confidence interval for the population mean. Assume the population is normally...
-
The Home Depot is the leading retailer in the home improvement industry and one of the 10largest retailers in the United States. The company included the following on its January 29, 2012, balance...
-
A chemist finds that the addition of (+)-epinephrine to the catalytic reduction of butan-2-one (Figure 5-16) gives a product that is slightly optically active, with a specific rotation of +0.45o...
-
1. Make a model of each compound, draw it in its most symmetric conformation, and determine whether it is capable of showing optical activity. (a) 1-bromo-1-chloroethane (b) 1-bromo-2-chloroethane...
-
Draw three-dimensional representations of the following compounds. Which have asymmetric carbon atoms? Which have no asymmetric carbons but are chiral anyway? Use your models for parts (a) through...
-
Three not-for-profit organizations operate separately in Sunland City. The most established entity, Sunland Support, has been around for over 100 years and generates by far the largest amount of...
-
E. Chrissy's Vases is a boutique selling artisan vases. The boutique purchases several styles of vases from one source. The cost of the vases to the boutique is $10 each. All other costs for...
-
Hampton Muffins sells its muffins to restaurants and coffee houses for an average selling price of $26 per case. The following information relates to the budget for Hampton Muffins for this year (all...

Study smarter with the SolutionInn App


