Identify the major and minor products for each of the following E2 reactions: a. b. c. d.
Question:
a.
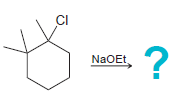
b.
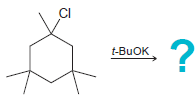
c.
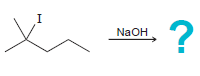
d.
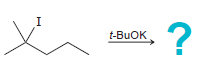
e.
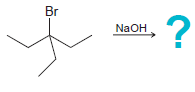
f.
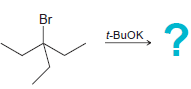
Transcribed Image Text:
CI NaOEt CI t-BUOK
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
a b c d e f ...View the full answer

Answered By

Anoop V
I have five years of experience in teaching and I have National Eligibility in teaching (UGC-NET) .
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the major and minor products for each of the following E1 reactions: a. b. c. d. H,SO. eat ETOH Heat Br
-
Identify the major and minor products for the E2 reaction that occurs when each of the following substrates is treated with a strong base: a. b. c. d. e. f. g. h. Br
-
Identify the major and minor products for the E2 reaction that occurs when each of the following substrates is treated with a strong base: a. b. c. d. e. f. g. h. Br
-
A 14-foot piece of string is cut into two pieces so that the longer piece is 2 feet longer than twice the shorter piece. Find the lengths of both pieces. What is the lenath of the shorter oiece?1...
-
The saturated density sat of a soil is 137 lbf/ft3. Find the buoyant density of this soil in both lbf/ft3 and kg/m3.?
-
Show, by a simple numerical example, that the covariance method may yield an AR system with poles outside the unit circle in the \(z\) plane.
-
What is the difference between null and alternative hypothesis?
-
Discuss the sequence in which the major components of the master budget are prepared. Why is it necessary to prepare the components in such a sequence?
-
If a project has an initial cost of $100,000 and it generates a positive cash flow of $17,000 a year for six years, what is the project internal rate of return?
-
A European apparel manufacturer has production facilities in Italy and China to serve its European market, where annual demand is for 1.9 million units. Demand is expected to stay at the same level...
-
Arrange each set of compounds in order of reactivity toward an E2 process: a. b. Br Br Br
-
How do narrow-spectrum pesticides differ from broad-spectrum pesticides?
-
What are some advantages and disadvantages of housing a project in a functional form?
-
ABC Veterinary Practice provides boarding services. For its "Regular Suite," it estimates its costs as follows: Variable cost per night (labor, food, electricity, nightly treat, etc.) = $15 Fixed...
-
Glory Enterprises quality control report for August contains the following items: Liability costs associated with defective products Disposal costs of defective products failing inspection Disposal...
-
Franklin Company deposits all cash receipts on the day they are received and makes all cash payments by check. At the close of business on August 31, its Cash account shows a debit balance of $23,662...
-
integral: Consider the region R defined by x + y 9. Compute the following Re e+y dA
-
The payroll register of Heritage Co . indicates $ 4 , 8 0 0 of social security withheld and $ 1 , 2 0 0 of Medicare tax withheld on total salaries of $ 8 0 , 0 0 0 for the period. Earnings of $ 1 4 ,...
-
A cotton ball dipped in alcohol is wiped across a tabletop. Would the resulting smell of the alcohol be more or less noticeable if the tabletop were much warmer? Explain.
-
If the cylinder described in Problem 21.3 were initially heated to 500F, how long would it take for the center of the cylinder to cool to 240F if it were constructed of a. Copper? b. Brass? c. Nickel?
-
Ketones react with acetylide ion (Section 8.7) to give alcohols. For example, the reaction of sodium acetylide with 2-hutanone yields 3-methyl-l-pentyn-3-ol: (a) Is the product chiral? Is it...
-
Imagine that another reaction similar to that in Problem 9.81 is carried out between sodium acetylide and (R)-2-phenylpropanal to yield 1-phenyl- 3-butyn-2-ol: (a) Is the product chiral? Is it...
-
Name the following compounds: CH C (a) H (b) CCCECHCH CCH CH CH (c) CH (d) CHH-3CH2H-CH CCH-CC3CCH3 H CH (9 HCHCHCHCCCH3 (e)
-
2. Consider the metal ion complex ferricyanide, [Fe(CN)6], obtained when potassium hexacyanoferrate(III) is dissolved in solution. a) Predict the number and intensity of all its ligand field (d-d)...
-
16) Chris, Bob, and Matt decide to help each other study during the final exams. Chris's favorite subject is chemistry, Bob loves biology, and Matt knows his math. Each studies his own subject as...
-
Match the column A with column B and Give Supported explanations. E Charge 1 Charge 2 Distance Apart Force Attracted/Repelled / Neither Trial 1 -4 C 4 cm Trial 2 -4 C -2 C 4 cm Trial 3 -4C -4C 4 cm...

Study smarter with the SolutionInn App


