Identify the major and minor products for the E2 reaction that occurs when each of the following
Question:
a.
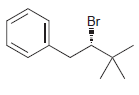
b.
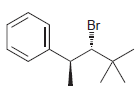
c.
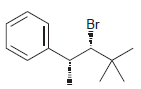
d.
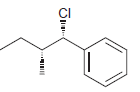
e.
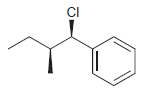
f.
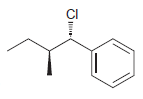
g.
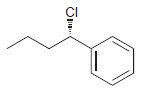
h.
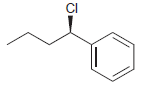
Transcribed Image Text:
Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (20 reviews)
a b c...View the full answer

Answered By

Kainat Shabbir
i am an experienced qualified expert with a long record of success helping clients overcome specific difficulties in information technology, business and arts greatly increasing their confidence in these topics. i am providing professional services in following concerns research papers, term papers, dissertation writing, book reports, biography writing, proofreading, editing, article critique, book review, coursework, c++, java, bootstarp, database.
5.00+
184+ Reviews
255+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When menthyl chloride is treated with a strong base, only one elimination product is observed. Yet, when neomenthyl chloride is treated with a strong base, two elimination products are observed. Draw...
-
When (1R, 2R)-2-bromocyclohexanol is treated with a strong base, an epoxide (cyclic ether) is formed. Suggest a mechanism for formation of the epoxide: Strong base Br An epoxide
-
Identify the major and minor products for each of the following E2 reactions: a. b. c. d. e. f. CI NaOEt CI t-BUOK
-
. What is the output of the following program? Write your answer in the box below. #include using namespace std; int main() { int a=4; int b=6; int i; int j; } int** k; i = &a; j = &b; *j = 8; if...
-
A sample of soil plus container weighs 397.6 g when the initial water content is 6.3%. The container weighs 258.7 g. How much water needs to be added to the original specimen if the water content is...
-
Commencing in 2007, foreign companies that were using IFRSs did not have to reconcile their reported profit to profit under U.S. GAAP . Access the 2011 and 2010 annual reports for Cenovus, a Canadian...
-
Determine the flowrate through the Venturi meter shown in Fig. P3.127 if ideal conditions exist. Figure P3.127 P = 735 kPa Q + 735 APU 31 mm P = 550 kPa 19 mm y=9.1 kN/m
-
Reliable Outdoor Furniture Company included the following stockholders equity on its year-end balance sheet at February 28, 2013: Requirements 1. Identify the different issues of stock that Reliable...
-
What health issue is prevalent in San Diego, California? What is the structure and function of public health at national, state, and local levels? what type of structure can be seen between levels of...
-
Refer to the data given in Case 5.44. Complete the same requirements, assuming that Home and Garden Products Ltd uses weighted average process costing. In calculating unit costs, round your answer to...
-
Show two different methods for preparing each of the following alkenes (one method using a sterically hindered base and the other method using a base that is not sterically hindered): a. b.
-
Identify the major and minor products for the E2 reaction that occurs when each of the following substrates is treated with a strong base: a. b. c. d. e. f. g. h. Br
-
Mansell Corporation faces stiff market competition. Top management is considering the replacement of its current production facility. The board of directors requires all capital investments to meet...
-
Kaitlin has $10,000 of savings that she may deposit with her local bank. Kaitlin wants to earn a real rate of return of at least 4 percent, and she is expecting inflation to be exactly 3 percent....
-
Zions nominal income will go up by 10 percent next year. Inflation is expected to be 2 percent next year. By approximately how much will Zions real income change next year? a. 2 percent b. 8 percent...
-
You are considering an investment in a clothes distributor. The company needs $109,000 today and expects to repay you $127,000 in a year from now. What is the IRR of this investment opportunity?...
-
What annual growth rate is needed for a country to double its output in 7 years? In 35 years? In 70 years? In 140 years?
-
For a balanced- \(\Delta\) load supplied by a balanced positive-sequence source, the line currents into the load are \(\sqrt{3}\) times the \(\Delta\)-load currents and lag by \(30^{\circ}\). (a)...
-
Differentiate the function. f (t) = 2t 3 - 3t 2 - 4t
-
Use the method of Example 4.29 to compute the indicated power of the matrix. 1 0 1
-
How would you prepare ds-2-butene starting from propyne, an alkyl halide, and any other reagents needed? This problem cant be worked in a single step. Youll have to carry out more than one reaction.
-
Beginning with 4-octyne as your only source of carbon, and using any inorganic reagents necessary, how would you synthesize the following compounds? (a) cis-4-Octene (b) Butanal (c) 4-Bromooctane (d)...
-
Beginning with acetylene and any alkyl halides needed, how would you synthesize the following compounds? (a) Decane (b) 2, 2-Dimethylhexane (c) Hexanal (d) 2-Heptanone
-
Braverman Company has two manufacturing departments-Finishing and Fabrication. The predetermined overhead rates in Finishing and Fabrication are $28.00 per direct labor-hour and 120% of direct...
-
A stock had returns of 28%, 1%, and 13% over the previous 3 years. The average return on this stock over the 3-year period was 14%. What was the variance of this stock's returns over the previous 3...
-
An airline will purchase 2 million gallons of jet fuel in one month and hedges using heating oil futures. From historical data, the standard deviation of heating oil futures contract is sF =0.031...

Study smarter with the SolutionInn App


