Identify whether each of the following compounds exhibits a molecular dipole moment. For compounds that do, indicate
Question:
a. CHCl3
b. CH3OCH3
c. NH3
d. CCl2Br2
e.
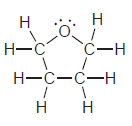
f.
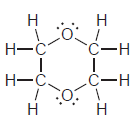
g.
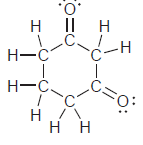
h.
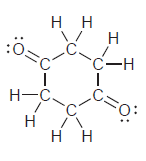
i.
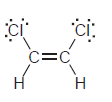
j.
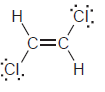
k.
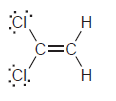
l.
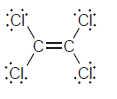
Transcribed Image Text:
Н Н б: Н—с С—н Н—с—с—н Н Н Н Н—с "С—н Н—С. С—н Н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
a b c d e ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify whether each of the following compounds is chiral or achiral: a. b. c. d. e. f. g. h. i. j. k. l. m. n. o. p. CI
-
Identify whether each of the following compounds can be made using a direct Friedel-Crafts alkylation or whether it is necessary to perform an acylation followed by a Clemmensen reduction to avoid...
-
Identify whether each of the following compounds is expected to be water soluble: (a) (b) (c) -NH2 -NH2
-
Find the lengths of the curves. The curve r = a sin 2 (/2), 0 , a > 0
-
Discuss the sustainability of economic growth. Is it possible for economic growth to continue forever? Why or why not? What are some influences on the rate of population growth? Can population...
-
In this exercise, we consider the amounts that are borrowed for single family home purchases in Las Vegas, Nevada, during 2010. Use the data file vegas5_small for this exercise. a. Compute summary...
-
Consider the fuel consumption data in Table B.18. Regressor \(x_{1}\) is an indicator variable. Perform a thorough analysis of these data. What conclusions do you draw from this analysis? y X2 X3 X4...
-
A car dealer acquires a used car for $ 14,000, terms FOB shipping point. Additional costs in obtaining and offering the car for sale include $ 250 for transportation-in, $ 900 for import duties, $...
-
Activity Optomistic Most Likely Pessimistic Critical A 4 6 9 no B 2 3 9 Yes C 12 14 15 YES D 10 28 29 NO E 3 4 5 Yes F 2 4 6 Yes What is the expected Project Completion time? ...
-
A vapor mixture having equal volumes of NH3 and N2 is to be contacted at 20?C and 1 atm (760 torr) with water to absorb a portion of the NH3. If 14 m3 of this mixture is brought into contact with 10...
-
Predict the value for the specific rotation of the following compound. Explain your answer. - , .
-
Which of the following compounds has the larger dipole moment? Explain your choice: CHCl 3 or CBrCl 3
-
Bluestone Metals, Inc., is a metal fabrication firm that manufactures prefabricated metal parts for customers in a variety of industries. The firms motto is If you need it, we can make it. The CEO of...
-
In the Opening Case at the beginning of the chapter, Superior Savings owned a properly executed and legally recorded mortgage on the building at 6802 Superior Avenue in Cleveland. The mortgagee,...
-
What is securitization and how did it contribute to the financial crisis?
-
How does the management of a limited liability company operate?
-
What are the different views of specific partnership property in partnership law?
-
Nike was founded in 1964 by Bill Bowerman. Located in Beaverton, Oregon, in the USA, the company was initially known as Blue Ribbon Sports. In 1972, it launched the first Nike shoe, but it was not...
-
What is the range of the cotangent function?
-
In Problems 1522, find the principal needed now to get each amount; that is, find the present value. To get $750 after 2 years at 2.5% compounded quarterly.
-
Write structures for the products of the following reactions: (a) C6H5CH2OH + C6H5N==C==O : (b) ClCOCl + excess CH3NH2 : (c) Glycine (H3+NCH2CO2-) + C6H5CH2OCOCI HO:- (d) Product of (c) + H2, Pd :...
-
Using decarboxylation reactions outline a synthesis of each of the following from appropriate starting materials: (a) 2-Hexanone (b) 2-Methylbutanoic acid (c) Cyclohexanone (d) Pentanoic acid
-
Diacyl peroxides decompose readily when heated. (a) What factor accounts for this instability? (b) The decomposition of a diacyl peroxide produces CO2. How is it formed? (c) Diacyl peroxides are...
-
If the local professional basketball team, the Sneakers, wins today's game, they have a 3 chance of winning their next game. If they lose this game, they have a chance of winning their next game. a)...
-
There is a function f of the form .12 f(x) = ax + x 13 for which f(0.1) = 6.06 10 and (0.9) = 0.03577. Determine a and B, and assess the sensitivity of these parameters to slight changes in the...
-
The order states: Give antihistamine elixir 2.5mg/kg/dose PO q4h. The patient weighs 143lbs. The drug is available as Antihistamine Elixir 200mg in every 8mL. Answer each of the following questions. ...

Study smarter with the SolutionInn App


