Identify which compound is expected to have a lower pK a . Justify your choice. N-H H.
Question:
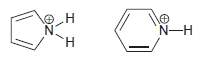
Transcribed Image Text:
N-H H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 91% (12 reviews)
The first compound is expected to be more ac...View the full answer

Answered By

Parvesh Kumar
I am an experienced Mathematics and Statistics tutor with 10 years of experience teaching students and working professionals. I love teaching students who are passionate to learn subjects or wants to understand any mathematics and statistics concept at graduation or master’s level. I have worked with thousands of students in my teaching career. I have helped students deal with difficult topics and subjects like Calculus, Algebra, Discrete Mathematics, Complex analysis, Graph theory, Hypothesis testing, Probability, Statistical Inference and more. After learning from me, students have found Mathematics and Statistics not dull but a fun subject. I can handle almost all curriculum of mathematics. I did B.Sc (mathematics), M.Sc (mathematics), M.Tech (IT) and am also Gate (CS) qualified. I have worked in various college and school and also provided online tutoring to American and Canadian students. I look forward to discussing with you and make learning a meaningful and purposeful
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each pair of compounds below, identify which compound is more acidic and explain your choice: (a)
-
For each pair of compounds, identify which compound is more acidic and explain your choice. (a) 2,4-Dimethyl-3,5-heptanedione or 4,4-Dimethyl-3,5-heptanedione (b) 1,2-Cyclopentanedione or...
-
Two compounds have the formula Pt(NH3)2Cl2. (Compound B is cisplatin). They have square planar structures. One is expected to have a dipole moment; the other is not. Which one would have a dipole...
-
At the beginning of compression in a diesel cycle, T = 540 R, P = 30 lbf/in. 2 , and the state after combustion (heat addition) is 2600 R and 1000 lbf/in. 2 . Find the compression ratio, the thermal...
-
On June 3, 2012, Irvine Corporation sells $45,000 of merchandise to a customer on account with terms of 2/10,n/30 Prepare the journal entries to: A) Record the sale using the gross method. B) Assume...
-
In the case in exercise 4, calculate the asymptotic wave function in the metastable region of time, and in the out region. Data From Exercise 4:- If we have two resonances (complex poles) for...
-
For each of the following situations, calculate the degrees of freedom \((d f)\), identify the critical values (assume \(\mathbf{a}=.05\) [two-tailed]), calculate the standard error of the mean \(s\)...
-
Dinos, Inc., makes a variety of T-shirts with logos. The company has discovered a new market for sweatshirts with logos. Market research indicates that a sweatshirt like this would sell well in the...
-
At the beginning of the year, Nowak Ltd . had the following amounts on its statement of financial position. Use this information to answer each of the following independent questions. s a ) Nowak s...
-
Presented below is a combined single-step income and retained earnings statement for Hardrock Mining Co. for 20X1. Statement of Income and Retained Earnings for the Year Ended December 31, 20X1 ($ in...
-
Go to the beginning of Section 18.1 where 8 best-selling drugs were shown. Review the structures of those compounds, and identify all of the aromatic rings that are not already highlighted in red. ...
-
Meclizine, shown below, is an antiemetic (prevents nausea and vomiting). a) Would you expect meclizine to be an antihistamine as well? Justify your answer. b) This drug is known to cause sedation....
-
You often see margins of errors in polling results listed as 3%. What sample size do you need to have a margin of error of at most 0.03 for a 95% confidence interval? To answer this question, set the...
-
Review The brick kitchen blog (https://www.thebrickkitchen.com/). The blog review should be a critical look and evaluation at the blog itself, rather than a summary of its content. The review should...
-
Please respond to one or more of the following: Blogs 1. If you are currently a blog reader, please pick a blog that you like to read on a regular basis. Give a brief description of the type of...
-
4. The Aerospaces Ltd. has manufacture two products with data as presented below: Annual overhead of Aerospace's Ltd. based on each cost pool as follow: Required: Calculate overhead cost per unit...
-
Jan exchanges stock that she owns in Rocky Corporation with a value of $21,500 for stock in Lucy Corporation. The stock that she received from Lucy had a value of $20,000 when received. In addition,...
-
Summative Assessment Recall 1 example of an effective social media post from a social media site or platform or from a blog site you have read and consider these questions as you prepare for this...
-
One card is selected at random from a deck of cards. Determine the probability that the card selected is A card greater than 3 and less than 8.
-
Gopher, Inc. developing its upcoming budgeted Costs of Quality (COQ) with the following information: Expense Item Budget Raw Materials Inspection $ 15,000 EPA Fine 200,000 Design Engineering 15,000...
-
Which SN1 reaction of each pair would you expect to take place more rapidly? Explain your answer. (a) (1) Or (2) (b) (1) Or (2) (c) (1) Or (2) (d) (1) Or (2) (e) (1) Or (2) OH + HCI HHBr Br + HCI +...
-
Show how you might use a nucleophilic substitution reaction of 1-bromopropane to synthesize each of the following compounds. (You may use any other compounds that are necessary.) (a) (b)...
-
With methyl, ethyl, or cyclopentyl halides as your organic starting materials and using any needed solvents or inorganic reagents, outline syntheses of each of the following. More than one step may...
-
Suppose you invest 52%, 28%, and 20% of your wealth into a stock, the market, and a risk-free asset, respectively. The beta of the stock is 1.1. What is the beta of the portfolio?
-
An investment of $21745, earning compound interest, grows by $2278 in one year. At this rate of growth, how long will it take for the original investment to double?
-
On September 12, 3,400 shares of Denver Company's common stock are acquired at a price of $58 per share plus a $170 brokerage commission. On October 15, an $1.20-per-share dividend was received on...

Study smarter with the SolutionInn App


